
(a)
Interpretation:
The systematic name of the compound
Concept Introduction:
Rules for naming compound:
- 1) The carbon atoms in the longest chain have to be counted.
- 2) The substituents and multiple bonds have to be identified and counted and the suffix “-ene” is added.
- 3) The backbone carbon atoms have to be numbered by assigning the lowest number from the starting end that contains the double bond.
Rules for naming compound:
- 1) The carbon atoms in the longest chain have to be counted.
- 2) The substituents and multiple bonds have to be identified and counted and the suffix “-yne” is added.
- 3) The backbone carbon atoms have to be numbered by assigning the lowest number from the starting end that contains the double bond.
Geometrical isomers: In geometrical isomers, atoms have different arrangements on either side of a double bond above or below the ring of a cycloalkane or cycloalkane. If the atoms are present on the same side of the double bond, then it is cis-isomer and if they are present on the opposite side of the double bond, then it is trans-isomer.
(a)
Answer to Problem 11.8E
The systematic name of the compound
Cis-
Explanation of Solution
The given compound is,
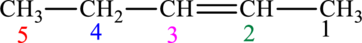
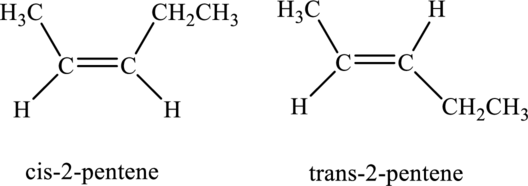
The compound is identified as alkene. The parent chain of the compound is pentane and a double bond is seen at the carbon second position. Hence, the systematic name of the compound is
Cis-
(b)
Interpretation:
The systematic name of the compound
Concept Introduction:
Refer to part (a).
(b)
Answer to Problem 11.8E
The systematic name of the compound
The geometrical isomers are not possible in
Explanation of Solution
The given compound is,
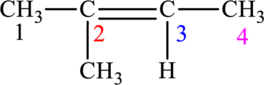
The compound is identified as alkene. The parent chain of the compound is butane. One methyl group is present in the carbon second position and a double bond is seen at the carbon second position. Hence, the systematic name of the compound is
No geometrical isomers are possible in
(c)
Interpretation:
The systematic name of the compound
Concept Introduction:
Refer to part (a).
(c)
Answer to Problem 11.8E
The systematic name of the compound
No geometrical isomers are possible in
Explanation of Solution
The given compound is,
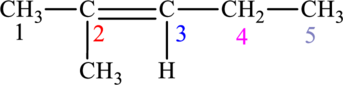
The compound is identified as alkene. The parent chain of the compound is pentane. One methyl group is present in the carbon second position and a double bond is seen at the carbon second position. Hence, the systematic name of the compound is
No geometrical isomers are possible in
(d)
Interpretation:
The systematic name of the compound
Concept Introduction:
Refer to part (a).
(d)
Answer to Problem 11.8E
The systematic name of the compound
No geometrical isomers are possible in
Explanation of Solution
The given compound is,
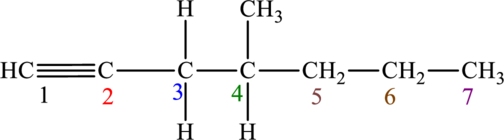
The compound is identified as alkyne. The parent chain of the compound is heptane. One triple bond is present in carbon first position and one methyl substituent is present in carbon fourth position. Hence, the systematic name of the compound is
Geometrical isomers are not possible because triple bond has only one substituent each.
(e)
Interpretation:
The systematic name of the compound
Concept Introduction:
Refer to part (a).
(e)
Answer to Problem 11.8E
The systematic name of the compound
No geometrical isomers are possible in
Explanation of Solution
The given compound is,
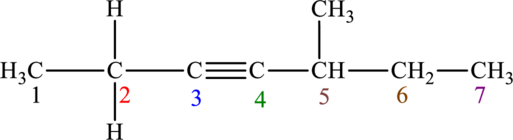
The compound is identified as alkyne. The parent chain of the compound is heptane. One triple bond is present in carbon third position and one methyl substituent is present in carbon fifth position. Hence, the systematic name of the compound is
Geometrical isomers are not possible because triple bond has only one substituent each.
Want to see more full solutions like this?
Chapter 11 Solutions
Chemical Principles: The Quest for Insight
- In a recent year, the United States produced 6.26 × 109kg1,2-dichloroethane and 3.73 × 109 kg vinyl chloride.Assuming that all significant quantities of vinyl chloridewere produced from 1,2-dichloroethane, what fraction ofthe 1,2-dichloroethane production went into making vinylchloride? What mass of hydrogen chloride was generatedas a by-product?arrow_forward5. How many isomers are there Of C6H14? 6. Aside from a halogenated alkane, what other product results from the bromination of hexane? Write the formulaarrow_forwardFor each of the following compounds,determine whether each is optically active. For optically active compounds, identify the chiral carbon: ethane, 2-chloro-2-methylbutane, CH3CH(NH2)COOH, CH3CH2CHClCH3and CH3CH2CH2CH3. And for non-optical active compounds, just provide the structure of the compoundarrow_forward
- Write the structural formula of a compound with an unbranched chain of five carbon atoms that is an alkane. You do not have to consider stereochemistry. Assume for the purposes of this problem that, to be an alcohol (-ol) or an amine (-amine), the hydroxyl or amino group must be bonded to a tetrahedral (sp3 hybridized) carbon atom. If more than one structure fits the description, draw them all. Ensure alkyne bonds are drawn linearly.arrow_forwardDetermine the molecular geometry and polarity of hydroxylamine (NH2OH), formaldehyde (CH2O), and formaldoxime (H2CNOH). Justify your deductions. Is there a difference between reactants and products for both features?.arrow_forwardDraw all possible isomers of heptane, C7H16, and give their corresponding names.arrow_forward
- C7H9N has 4 degrees of unsaturation, what are the possible structures?arrow_forwardOne mole of an unknown hydrocarbon, compound C, in the presence of a platinum catalyst, adds 98.9 L of hydrogen, measured at 744 mm Hg and 22 degrees C , to form a saturated alkane which contains one ring. When one mole of compound C is reacted with ozone, followed by reduction with (CH3)2S , four moles of only one product was formed, whose condensed molecular formula is CHO -CHO. Give the structure of compound C. Explain your reasoningarrow_forwardAn unknown hydrocarbon Q has a formula C6H12. Q Reacts with osmium tetroxide to give a diol R. When oxidized with KMnO4 in an acidic medium, Q gives two products. One product is propanoic acid and the other a ketone S. Provide reaction equations to identify the possible structures of Q, R and S.arrow_forward
- The carbon–carbon bond length in C2H2 is 1.20 Å, that in C2H4 is 1.34 Å, and that in C2H6 is 1.53 Å. Near which of these values would you predict the bond length of C2 to lie? Is the experimentally observed value, 1.31 Å, consistent with your prediction?arrow_forwardThe sex attractant of the female tiger moth is an alkane of molecular formula C 18H 38. Is this molecule an acyclic alkane or a cycloalkane?arrow_forwardWhat is the degree of unsaturation and structure for C8H5NO2 ?arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

