
Biochemistry: The Molecular Basis of Life
6th Edition
ISBN: 9780190209896
Author: Trudy McKee, James R. McKee
Publisher: Oxford University Press
expand_more
expand_more
format_list_bulleted
Question
Chapter 12, Problem 24RQ
Summary Introduction
To review:
The given gluco-cerebroside, and the bond in it that is cleaved by glucocerebrosidase.
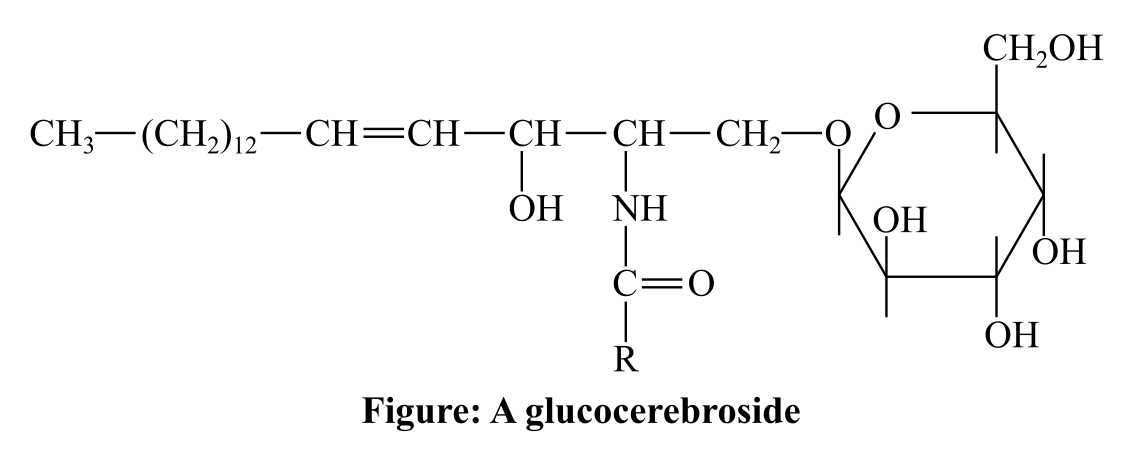
Introduction:
Cerebroside refers to monoglycosylceramides, which is an essential part of neuron and muscle cell. In these compounds, a ceramide group having a sugar residue at the 1-hydroxyl moiety is present. These are of two types, namely, glucocerebroside and glycerocerebroside.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is proteorhodopsin and why is it so named?
what is the role of amino acids in purine metabolism?
What is ornithine transcarbamylase (OTC)deficiency ?
Chapter 12 Solutions
Biochemistry: The Molecular Basis of Life
Ch. 12 - Prob. 1QCh. 12 - Prob. 2QCh. 12 - Prob. 3QCh. 12 - Prob. 4QCh. 12 - Prob. 5QCh. 12 - Prob. 6QCh. 12 - Prob. 7QCh. 12 - Prob. 8QCh. 12 - Prob. 9QCh. 12 - Prob. 10Q
Ch. 12 - Prob. 1RQCh. 12 - Prob. 2RQCh. 12 - Prob. 3RQCh. 12 - Prob. 4RQCh. 12 - Prob. 5RQCh. 12 - Prob. 6RQCh. 12 - Prob. 7RQCh. 12 - Prob. 8RQCh. 12 - Prob. 9RQCh. 12 - Prob. 10RQCh. 12 - Prob. 11RQCh. 12 - Prob. 12RQCh. 12 - Prob. 13RQCh. 12 - Prob. 14RQCh. 12 - Prob. 15RQCh. 12 - Prob. 16RQCh. 12 - Prob. 17RQCh. 12 - Prob. 18RQCh. 12 - Prob. 19RQCh. 12 - Prob. 20RQCh. 12 - Prob. 21RQCh. 12 - Prob. 22RQCh. 12 - Prob. 23RQCh. 12 - Prob. 24RQCh. 12 - Prob. 25RQCh. 12 - Prob. 26RQCh. 12 - Prob. 27RQCh. 12 - Prob. 28RQCh. 12 - Prob. 29RQCh. 12 - Prob. 30RQCh. 12 - Prob. 31RQCh. 12 - Prob. 32RQCh. 12 - Prob. 33RQCh. 12 - Prob. 34RQCh. 12 - Prob. 35RQCh. 12 - Prob. 36RQCh. 12 - Prob. 37RQCh. 12 - Prob. 38RQCh. 12 - Prob. 39RQCh. 12 - Prob. 40RQCh. 12 - Prob. 41RQCh. 12 - Prob. 42RQCh. 12 - Prob. 43RQCh. 12 - Prob. 44RQCh. 12 - Prob. 45RQCh. 12 - Prob. 46FBCh. 12 - Prob. 47FBCh. 12 - Prob. 48FBCh. 12 - Prob. 49FBCh. 12 - Prob. 50FBCh. 12 - Prob. 51FBCh. 12 - Prob. 52FBCh. 12 - Prob. 53FBCh. 12 - Prob. 54FBCh. 12 - Prob. 55FBCh. 12 - Prob. 56SACh. 12 - Prob. 57SACh. 12 - Prob. 58SACh. 12 - Prob. 59SACh. 12 - Prob. 60SACh. 12 - Prob. 61TQCh. 12 - Prob. 62TQCh. 12 - Prob. 64TQCh. 12 - Prob. 65TQCh. 12 - Prob. 66TQCh. 12 - Prob. 67TQCh. 12 - Prob. 68TQCh. 12 - Prob. 71TQCh. 12 - Prob. 72TQ
Knowledge Booster
Similar questions
- Why is phenylketonuria resulting from dihydropteridine reductase deficiency a more serious disorder than PKU resulting from phenylalaninehydroxylase deficiency?arrow_forwardWhy is oxaloacetate an intermediate in gluconeogenesis?arrow_forwardWhat form of activated glucose is used in the biosynthesis ofglycogen by bacteria?arrow_forward
- Why does the enzyme reaction for chymotrypsin proceed in two phases?arrow_forwardThis is a conjectural question: If the reactive part of coenzyme A is the thioester, why is the molecule socomplicated?arrow_forwardPKU is an abbreviation for what disorder? What are the symptoms of PKU? How can PKU be treated for a nearly normal life?arrow_forward
- What is a toxic phospholipase?arrow_forwardGive a detailed account of the tricarboxylic acid pathway and how it is regulated in eukaryotic cells. could you please help me to write 700 word essay on this topic with examples. many thanksarrow_forwardtrue or false: Guanylate phosphorylase is responsible for converting guanosine monophosphate into guanosine diphosphatearrow_forward
- what is the difference between catabolic and anabolicarrow_forwardTrue or False. The Krebs cycle is considered to be an anabolic pathway because some of the reactions are considered reversible.arrow_forwardQUESTION 26 During gluconeogenesis, whereby liver cells convert pyruvate to glucose, Fructose-6-phosphate (F6P) is converted to Glucose-6-phosphate (G6P). If the standard equilibrium concentrations are: [F6P] = 0.52 M and [G6P] = 1.48 M, then Keq’ is ______ and the reaction is ________. Fructose-6-P ó Glucose-6-P > 1; exergonic > 1; endergonic < 1; exergonic < 1; endergonicarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON

Biochemistry
Biochemistry
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:9781464126116
Author:David L. Nelson, Michael M. Cox
Publisher:W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul...
Biochemistry
ISBN:9781118918401
Author:Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:WILEY

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Fundamentals of General, Organic, and Biological ...
Biochemistry
ISBN:9780134015187
Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:PEARSON