
Concept explainers
Interpretation:
The reason should be explained for the formation of the unexpected product 2-methyl-
2-pentene on dehydration of 4-methyl-2-pentanol.
Concept Introduction:
Dehydration reaction:
Removal of water molecule from the reaction when the alcohol is treated with strong acid like sulfuric acid.
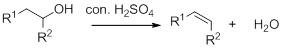
Alcohol is reaction with concentrated sulfuric acid, first alcohol gets protonated forms carbocation (more stable carbocation) followed by elimination of proton (
Tertiary carbocation is more stable than the secondary, secondary carbocation is more stable than primary.

In dehydration reaction, sulfuric acid is act as a proton donor, and which is used to protonate the alcohol and makes carbocation therefore sulfuric acid is the driving force of the reaction.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
Fundamentals Of General, Organic, And Biological Chemistry Volume 1 Second Custom Edition For Washington State University, 2/e
- Is D-2-deoxygalactose the same chemical as D-2-deoxyglucose? Explain.arrow_forwardRefer back to Figure 4.9. What type of isomers areacetone and propanal? How many asymmetric carbons are present in aceticacid, glycine, and glycerol phosphate? Can these three molecules exist asforms that are enantiomers?arrow_forwardWhat is the empirical formula for C3H6O3? C3H6O3 C6H12O6 CH2O None of thesearrow_forward
- Is D-2-deoxygalactose the same chemical as D-2-deoxyglucose? Draw and explainarrow_forwardIf the phosphorus atom in 3-phosphoglycerate is radioactively labeled, where will the label be when the reaction that forms 2-phosphoglycerate is over?arrow_forwardIt is speculated that, in 1954, the scientist Allan Turing (who was instrumental in decoding the German Enigma code during the Second World War) ate an apple, which had been injected with a solution of cyanide, to commit suicide. How does cyanide poisoning work on a molecular level, and why can this type of toxic effect not be prevented as in the case of methanol poisoning?arrow_forward
- Name the following fatty acids by the symbol (C:B)arrow_forwardWhat would be the fate of oxaloacetate if there were sufficient fluoroacetate present?arrow_forwardWhat product is formed when a solution of A and B is treated with mild base? This reaction is the first step in the synthesis of rosuvastatin (sold as a calcium salt under the trade name Crestor), a drug used to treat patients with high cholesterol.arrow_forward
- The metabolic intermediate acetyl phosphate is an anhydride formed from acetic acid and phosphoric acid. What is the structure of acetyl phosphate?arrow_forwardIs cholesterol the only compound in egg yolks that is soluble in 2-propanol? why or why not?arrow_forwardExplain the process of polyacrylamide reaction, and what are the two reagents are crucial for this reaction?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





