
a)
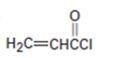
Interpretation:
Whether the compound shown will be a good dienophile or not is to be predicted.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond.
To predict:
Whether the compound shown will be a good dienophile or not.
b)
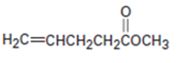
Interpretation:
Whether the compound shown will be a good dienophile or not is to be predicted.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond.
To predict:
Whether the compound shown will be a good dienophile or not.
c)
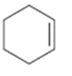
Interpretation:
Whether the compound shown will be a good dienophile or not is to be predicted.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond.
To predict:
Whether the compound shown will be a good dienophile or not.
d)
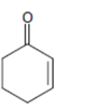
Interpretation:
Whether the compound shown will be a good dienophile or not is to be predicted.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond.
To predict:
Whether the compound shown will be a good dienophile or not.
e)
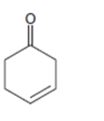
Interpretation:
Whether the compound shown will be a good dienophile or not is to be predicted.
Concept introduction:
In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction takes place through 1,4 addition of the dienophile into the diene through a cyclic transition state. These reactions occur rapidly with dienophiles having electron withdrawing substituent groups in conjugation with the double bond.
To predict:
Whether the compound shown will be a good dienophile or not.
Trending nowThis is a popular solution!

Chapter 14 Solutions
Organic Chemistry
- Predict mechanistically what stereoisomers of 3-chloro-5-methylcyclohexene should form when (image below) is treated with Lucas reagent (ZnCl2/H30+Cl-).arrow_forwardWhich of the following conjugated dienes will not react with a dienophile in a Diels–Alder reaction?arrow_forwardConsider the following dienes in a Diels–Alder reaction with ethene. Which will react the fastest? Which will react the slowest? Explainarrow_forward
- Write the products of the reaction below and indicate if the product is R or S or racemic or achiral.arrow_forwardWhat would be the major and minor alkenes formed from the E2-elimination of cis-1? Thank you :)arrow_forwardWhich dienophile in each pair is more reactive in a Diels–Alder reaction?arrow_forward
- If this diene were to be treated with HBr at -10C, where would the Br attach to the structure in the major product?arrow_forwardWhat is the product (include the stereochemistry) of the 6 pi electron electrocyclic reaction below, use the appropriate Woodward-Hoffman rule.arrow_forwardPlease identify priorities of functional groups and name following molecules based on thier stereochemistry as R or Sarrow_forward
