
Concept explainers
(a)
Interpretation:
Structure of the 2-octanone should be drawn.
Concept Introduction:
(b)
Interpretation:
The product should be identified by the reaction of 2-octanone with
Concept Introduction:
Addition of H2gas to a multiple bond is called hydrogenation. Aldehydes and ketones which are considered as carbonyl compounds, undergo reduction reactions in the presence of
In below diagram R is an alkyl group in ketone and
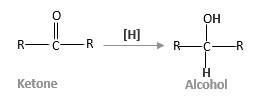
(c)
Interpretation:
Chirality center of the product which gain by the reaction of 2-octanone with
Concept Introduction:
Chirality is the presence of an asymmetric carbon center in a molecule and a molecule which contains a chiral center cannot superimpose on its mirror image.
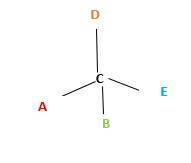
In the above diagram, where C is the chiral center/ asymmetric carbon center.
A, B, D, E are four different
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- Many insects utilize cyclic ketal structures as pheromones such as the structure shown below. The biosynthetic pathway involves the cyclization of this acetal from the straight chain structure. Draw the straight chain structure that could be used to form this acetal. Use wedges and dashes to correctly depict the stereochemistry. H3C- >arrow_forwardIdentify the functional groups in two drugs, atenolol and donepezil. Atenololis a β (beta) blocker, a drug used to treat hypertension (high bloodpressure), and donepezil (trade name Aricept) is used to treat mild tomoderate dementia associated with Alzheimer's disease.arrow_forwardDraw the products formed when each alcohol is oxidized with K 2Cr 2O 7. In some cases, no reaction occurs.arrow_forward
- which compound has the highest boiling point: hexan-2-ol or hexane-1,5-diol?arrow_forwardDraw the structure of (R) 3-hydroxy-4-cyclohexenone. Give the IUPAC name of this compound, including stereochemistry. CH₂ HỌ.Harrow_forwardCompound X and compound Y are constitutional isomers with the molecular formula C5H10. Compound X possesses a carbon-carbon double bond in the trans configuration, while compound Y possesses a carbon-carbon double bond that is not stereoisomeric: Draw the structure of compound X. Draw Your Solutionarrow_forward
- Draw the structure of 4-phenylbutanal, g-phenylbutyraldehyde:arrow_forwardWhat product is formed when the alcohol is oxidized with K2Cr2O7? In some cases, no reaction occurs (if so, draw the given alcohol).arrow_forward17) This compound is the product of: O II (CH3)2CHCOCH₂CH(CH₂CH3)2 A) the oxidation of a primary alcohol B) the reaction of a carboxylic acid and an alcohol C) the reaction of a carboxylic acid and ammonia D) the reaction of a ketone and an alcohol 0arrow_forward
- 6. Draw the structures of the products A, B, C and D. OH N2OH TSCI (S,2) B Pyridine (R)-2-Butanol TSCI LICI D Pyridine *OHarrow_forwardIn an advanced synthetic chemistry experiment, a researcher prepares a compound, ZY-7, by reacting a ketone (C5H100) with hydroxylamine (NH2OH), followed by heating in the presence of an acid catalyst. The resulting compound, ZY-7, is then treated with a solution of sodium nitrite (NaNO2) and hydrochloric acid (HCI) at low temperature. Identify the class of compound that ZY-7 most likely belongs to after this series of reactions." A) Amide B) Oxime C) Nitro compound D) Diazonium salt E) Ester Don't use chatgpt please provide valuable answerarrow_forwardWhat functional group/s is/are present in the structure? OH Br CH,- C- CH =CH -C- CH =CH – CHO CI double bond, carbonyl, hydroxyl double bond, carboxyl, hydroxyl double bond, carboxyl, halide, hydroxyl double bond, formyl, halide, hydroxyl double bond, formyl, hydroxylarrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning





