
Concept explainers
(a)
Interpretation:
The product formed in the reaction of
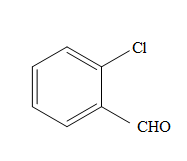
Concept Introduction:
Potassium dichromate is a good oxidizing agent and it has the tendency to oxidize alcohols to
(b)
Interpretation:
The product formed in the reaction of y13
Y13
Concept Introduction:
Potassium dichromate is a good oxidizing agent and it has the tendency to oxidize alcohols to aldehydes and aldehydes to carboxylic acids as well. An oxidizing agent is a chemical species which has the tendency to oxidize other to possible higher oxidation states by making them loose electron and thus itself gets reduced.
(c)
Interpretation:
The product formed in the reaction of
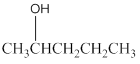
Concept Introduction:
Potassium dichromate is a good oxidizing agent and it has the tendency to oxidize alcohols to aldehydes and aldehydes to carboxylic acids as well. An oxidizing agent is a chemical species which has the tendency to oxidize other to possible higher oxidation states by making them loose electron and thus itself gets reduced.
(d)
Interpretation:
The product formed in the reaction of
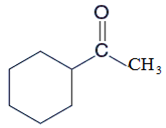
Concept Introduction:
Potassium dichromate is a good oxidizing agent and it has the tendency to oxidize alcohols to aldehydes and aldehydes to carboxylic acids as well. An oxidizing agent is a chemical species which has the tendency to oxidize other to possible higher oxidation states by making them loose electron and thus itself gets reduced.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- Classify each alkyl halide as 1°, 2°, or 3°. CH3 c. CHg-C-CHCH3 ČH3 ČI CH;CH2CH,CH,CH2-Br b. d. a.arrow_forward18. Ketone reduction Dicyclohexyl ketone Reduce the ketone. 1. NaBH4, ethanol 2. H3O+ H OH Dicyclohexylmethanol (88%) (a 2° alcohol)arrow_forwardDetermine the products of each carboxylic acid reaction. a. CH3-CH2-CH2-CH,-C-OH + H,SO, CH, — CH,— он HO. b. CH2 heat CH2 C-OH CH2arrow_forward
- Dehydration of alcohols can be done by using: O Fe2O3 O Cao O all of these O conc. H2SO4arrow_forward• Whał arc the IUPAC namar of the ff. Carboxylic acias? a. COOH COOH CH3 b. I f. .COOH CHJCH2 ÇHCHCOOH CH3 NO2 tON C. COOH ноос d. COOHarrow_forwardWhat is the major organic product obtained from the following reaction? A. B. OH -OH H₂SO4 C. D. Harrow_forward
- What is the major organic product obtained from the following reaction? A. B. OH OH 1.2 moles 2. H3O* C. MgBr هذه معه OHarrow_forwardDraw the products of each reaction. OH он CH3 Cl2 FeCla Cl2 a. b. С. FeCl,arrow_forwardWhat is the product of the following reaction? A. B. شما NH NH2Et, cat H* C. ZI avant HO. D. `N OHarrow_forward
- Which alkyl halide has the highest boiling point? A. CH3BrB. CH3FC. CH3ClD. CH3larrow_forwardtaken in order to gor the product Please explain the mechanisms/stepsarrow_forwardDraw the organic product(s) formed when CH3CH2CH2OH is treated with each reagent. a.H2SO4 b.NaH c.HCl + ZnCl2 d.HBr e.SOCl2, pyridine f.PBr3 g.TsCl, pyridine h. [1] NaH; [2] CH3CH2Br [1] i.TsCl, pyridine; [2] NaSH j.POCl3, pyridinearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





