
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
3rd Edition
ISBN: 9781259298424
Author: SMITH
Publisher: VST
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 16, Problem 16.72P
Interpretation Introduction
(a)
Interpretation:
The acetal in salicin should be labeled.
Concept Introduction:
Addition of one molecule of alcohol to an
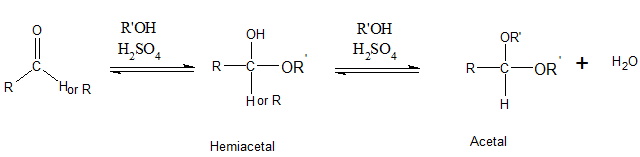
Interpretation Introduction
(b)
Interpretation:
Number of primary OH groups salicin contains should be determined.
Concept Introduction:
A primary OH group is a hydroxyl group which is attached to a primary C atom. Primary C atom has two H atoms attached to it along with one alkyl/aryl group.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Dexamethasone is a halogen-containing steroid used to treat infl ammation in rheumatoid arthritis and other conditions. (a) Classify the alkyl halide in dexamethasone as 1 °, 2 °, or 3 °. (b) Classify the hydroxyl groups as 1 °, 2 °, or 3 °.
Identify the three acetals in digoxin, a naturally occurring drug prescribed for patients with congestive heart failure and other heart ailments.
A: aromatic compound
B:
4
C:
N
X
28
N
0
C
Chapter 16 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
Ch. 16.1 - Prob. 16.1PCh. 16.1 - Draw the structure of the three constitutional...Ch. 16.2 - Prob. 16.3PCh. 16.2 - Give the structure corresponding to each TUPAC...Ch. 16.2 - Prob. 16.5PCh. 16.2 - Prob. 16.6PCh. 16.2 - Give the structure corresponding to each name. a....Ch. 16.3 - Which compound in each pair has the higher boiling...Ch. 16.3 - Acetone and progesterone are two ketones that...Ch. 16.4 - Prob. 16.10P
Ch. 16.5 - What product is formed when each carbonyl compound...Ch. 16.5 - Prob. 16.12PCh. 16.5 - Prob. 16.13PCh. 16.6 - What alcohol is formed when each compound is...Ch. 16.6 - Prob. 16.15PCh. 16.6 - Prob. 16.16PCh. 16.7 - Prob. 16.17PCh. 16.7 - Prob. 16.18PCh. 16.8 - Prob. 16.19PCh. 16.8 - Prob. 16.20PCh. 16.8 - Prob. 16.21PCh. 16.8 - Label the three acetals in solanine, the toxic...Ch. 16.8 - Prob. 16.23PCh. 16.8 - Prob. 16.24PCh. 16 - Prob. 16.25PCh. 16 - Prob. 16.26PCh. 16 - Prob. 16.27PCh. 16 - Prob. 16.28PCh. 16 - Prob. 16.29PCh. 16 - Prob. 16.30PCh. 16 - Prob. 16.31PCh. 16 - Prob. 16.32PCh. 16 - Prob. 16.33PCh. 16 - Prob. 16.34PCh. 16 - Give an acceptable name for each ketone. a. b. c....Ch. 16 - Prob. 16.36PCh. 16 - Prob. 16.37PCh. 16 - Draw the structure corresponding to each name. a....Ch. 16 - Prob. 16.39PCh. 16 - Prob. 16.40PCh. 16 - Prob. 16.41PCh. 16 - Prob. 16.42PCh. 16 - Draw out the structure of benzaldehyde, including...Ch. 16 - Prob. 16.44PCh. 16 - Which compound in each pair has the higher boiling...Ch. 16 - Prob. 16.46PCh. 16 - Prob. 16.47PCh. 16 - Prob. 16.48PCh. 16 - Prob. 16.49PCh. 16 - Prob. 16.50PCh. 16 - Prob. 16.51PCh. 16 - Prob. 16.52PCh. 16 - Prob. 16.53PCh. 16 - Prob. 16.54PCh. 16 - Prob. 16.55PCh. 16 - Consider the following ball-and-stick model....Ch. 16 - Prob. 16.57PCh. 16 - Prob. 16.58PCh. 16 - Prob. 16.59PCh. 16 - Prob. 16.60PCh. 16 - Prob. 16.61PCh. 16 - Prob. 16.62PCh. 16 - Prob. 16.63PCh. 16 - Prob. 16.64PCh. 16 - Prob. 16.65PCh. 16 - Prob. 16.66PCh. 16 - Prob. 16.67PCh. 16 - Prob. 16.68PCh. 16 - Prob. 16.69PCh. 16 - Label the functional group(s) in each compound as...Ch. 16 - Prob. 16.71PCh. 16 - Prob. 16.72PCh. 16 - What acetal is formed when each aldehyde or ketone...Ch. 16 - What acetal is formed when each aldehyde or ketone...Ch. 16 - Prob. 16.75PCh. 16 - Prob. 16.76PCh. 16 - Prob. 16.77PCh. 16 - Prob. 16.78PCh. 16 - Prob. 16.79PCh. 16 - Prob. 16.80PCh. 16 - Prob. 16.81PCh. 16 - Prob. 16.82PCh. 16 - Prob. 16.83PCh. 16 - Answer each question about phenylacetaldehyde,...Ch. 16 - Prob. 16.85PCh. 16 - Prob. 16.86PCh. 16 - Prob. 16.87PCh. 16 - Prob. 16.88PCh. 16 - Three constitutional isomers of molecular formula...Ch. 16 - Identify A—C in the following reaction sequenceCh. 16 - Androsterone is a male sex hormone that controls...Ch. 16 - Prob. 16.92PCh. 16 - Prob. 16.93PCh. 16 - Prob. 16.94PCh. 16 - Prob. 16.95PCh. 16 - Paraldehyde, a hypnotic and sedative once commonly...Ch. 16 - Prob. 16.97PCh. 16 - Prob. 16.98PCh. 16 - Prob. 16.99CPCh. 16 - Prob. 16.100CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Indinavir (trade name Crixivan) is a drug used to treat HIV. (a) At which sites can indinavir hydrogen bond to another molecule like itself? (b) At which sites can indinavir hydrogen bond to water?arrow_forwardWhy is it safe for us to consume foods like vinegar that contain acetic acids?arrow_forwardFenfluramine and phentermine are two components of fen–phen, an appetite suppressant withdrawn from the market in 1997 after it was shown to damage the heart valves in some patients. What products are formed when fenfluramine and phentermine are each treated with acetic acid (CH3CO2H)?arrow_forward
- Give the IUPAC name for each nitrile.arrow_forwardAspirin is the common name for the compound acetylsalicylic acid, widely used as a fever reducer and as a pain killer. Salicylic acid, whose name comes from Salix, the willow family of plants, was derived from willow bark extracts. In folk medicine, willow bark teas were used as headache remedies and other tonics. Nowadays, salicylic acid is administered in the form of aspirin which is less irritating to the stomach than salicylic acid. To prepare aspirin, salicylic acid is reacted with an excess of acetic anhydride. A small amount of a strong acid is used as a catalyst which speeds up the reaction. In this experiment, phosphoric acid will be used as the catalyst. The excess acetic acid will be quenched with the addition of water. The aspirin product is not very soluble in water so the aspirin product will precipitate when water is added. The synthesis reaction of aspirin is shown below: Actic anhydride 5 ml. Acetic acid Salicylic acid 28 Acetylsalicylie acid Procedure 1) Mix salicylic…arrow_forwardEtoposide, sold as a phosphate derivative with the trade name of Etopophos, is used for the treatment of lung cancer, testicular cancer, and lymphomas. (a) Locate the acetals in etoposide. (b) What products are formed when all of the acetals are hydrolyzed with aqueous acid?arrow_forward
- How many kinds of chemically non-equivalent carbons are there in each of the following compounds? a b علم CH₂-CH₂- +NH₂ OH CH₂ Aspirin (analgesic, antipyretic and antibiotic) The number of chemically non-equivalent carbons is The number of chemically non-equivalent carbons isarrow_forward7) I's the following compound a hydrate, hemiacetal, acetal, hemiketal or ketal? في المساجد LOCHarrow_forwardDraw an example of a lactone and lactam, label each.arrow_forward
- 2. What carboxylic acid and alcohol are needed to prepare each ester by Fischer esterification? a) b) c)arrow_forwardDraw 3,3-dichloropentanalarrow_forwardBarbiturates with rapid onset and short duration are: O used to treat epilepsy. O not used medically. O used as anesthetics. less addictive.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY