
(a)
Interpretation:
The
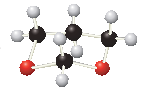
Concept introduction:
Ethers are derived from alcohols. The hydrogen in the hydroxyl group is replaced with an organic fragment in ethers.
Acetals are formed when alcohols are added to carbonyl groups. Acetals contain two ether groups.
The general formula for ethers and acetals is represented as follows:
(b)
Interpretation:
The functional group/s in the given compound needs to be identified as ether or acetal.
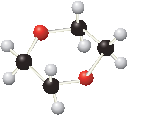
Concept introduction:
Ethers are derived from alcohols. The hydrogen in the hydroxyl group is replaced with an organic fragment in ethers.
Acetals are formed when alcohols are added to carbonyl groups. Acetals contain two ether groups.
The general formula for ethers and acetals is represented as follows:
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- Assign the IUPAC name to each of the following ethers. Name the smaller alkyl group as the alkoxy group. a. CH3CH2OCH2CH2CH3 b. c. d.arrow_forwardList the following compounds in order of increasing water solubility: a.ethoxyethane b.propanoic acid c.pentane d.1 butanolarrow_forwardPart: 1 / 2 Part 2 of 2 Provide the IUPAC name for this compound. X CH₂-CH₂-CH₂-COOH 5arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,  Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





