
(a)
Interpretation:
An acceptable name should be determined for the following acid:
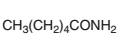
Concept introduction:
There are two acceptable names for a compound.
- IUPAC name
- Common name
Common names use older names of the compounds other than the prefix for carbon skeleton. For example, 'meth' prefix is 'form' as in methanamide is called formamide. For amides, 'ic acid' ending of the acid name is replaced with suffix 'amide'.
(b)
Interpretation:
An acceptable name should be determined for the following acid:
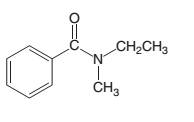
Concept introduction:
There are two acceptable names for a compound.
- IUPAC name
- Common name
Common names use older names of the compounds other than the prefix for carbon skeleton. For example, 'meth' prefix is 'form' as in methanamide is called formamide. For amides, 'ic acid' ending of the acid name is replaced with suffix 'amide'.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- What amine is formed by reduction of each amide?arrow_forwardThe presence of amides in living organisms is beneficial due its stability which results from being the least reactive carboxylic acid derivative. True or Falsearrow_forwardWhat products are formed when each amide is hydrolyzed with water and HCl?arrow_forward
- What ammonium salt is formed when each amine is treated with HCl? Draw the structure of the resulting salt.arrow_forwardClassify the specified amides as 1º, 2º, or 3º. Part 1 Select the single best answer. Part 2 Oxytocin, sold under the trade name Pitocin, is a naturally occurring hormone used to stimulate uterine contractions and induce labor. Classify the specified amides in oxytocin as 1º, 2º, or 3º.arrow_forwardWhat carbonyl compound and amine are formed by the hydrolysis of each compound?arrow_forward
- Select the name for each amide. ye N-ethylpropanamide N-methylbutanamide N-butylethanamide N-ethylbutanamide 4-cyclopentylbutanamide 5-cyclopentylpentanamide O cyclopentylbutanamide butanamide cyclopentane N-butylethanamide N-diethylacetamide N,N-diethylethanamide N,N-dimethylacetamide N-ethyl.N-methylbenzoate ON-ethyl,N-methyl-2-phenylpropanamide N-ethyl-N-methyl-2-phenylethanamide N,N-diethylbenzoatearrow_forwardAcid Anhydride Amine Thiol Peroxide Nitrile Arenearrow_forwardWhich compound makes the strongest amine base? `N CH3 -N- 1 2 3 4 Select an answer and submit. For keyboard navigation, use the up/down arrow keys to select an answer. a Compound 1 b Compound 2 Compound 3 d Compound 4arrow_forward
- explain why an N,N-disubstituted amide is less acidic than an ester.arrow_forwardDraw the structural formula for each amides. - N-Cycloclohexylacetamide - Benzamidearrow_forwardLabel each nitrogen-containing functional group in lidocaine, a local anesthetic, as an amine or amide, and classify it as 1 °, 2 °, or 3 °.arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
