
(a)
Interpretation:
The amide formed when butanoic acid (CH3CH2CH2CO2H) is heated with ammonianeeds to be determined.
Concept Introduction:
When
General reaction is represented as follows:
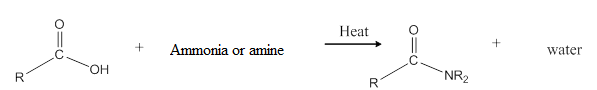
(b)
Interpretation:
The amide formed when butanoic acid (CH3CH2CH2CO2H) is heated with CH3CH2NH2 needs to be determined.
Concept Introduction:
When carboxylic acid is heated with an ammonia or an amine, amide formation can be observed.
General reaction is represented as follows:
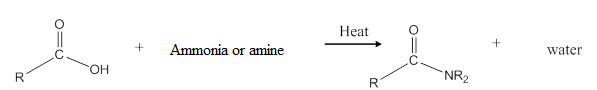
(c)
Interpretation:
The amide formed when butanoic acid (CH3CH2CH2CO2H) is heated with (CH3CH2)2NH needs to be determined.
Concept Introduction:
When carboxylic acid is heated with an ammonia or an amine, amide formation can be observed.
General reaction is represented as follows:
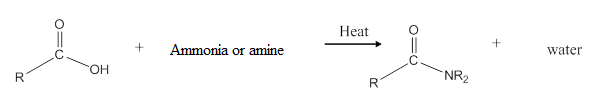
(d)
Interpretation:
The amide formed when butanoic acid (CH3CH2CH2CO2H) is heated with CH3NHCH2CH3 needs to be determined.
Concept Introduction:
When carboxylic acid is heated with an ammonia or an amine, amide formation can be observed.
General reaction is represented as follows:
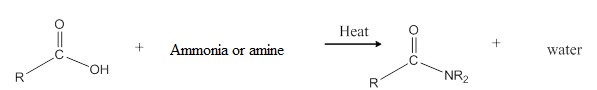
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- Draw the amide formed when 1-methylethylamine (CH3CH(CH3)NH2) is heated with each carboxylic acid.arrow_forwardWhat is the IUPAC name of a benzene ring with an amine group (NH2-) attached at carbon 1 and a bromine attached at carbon 2? O 2-bromoaniline O 2-bomotoluene O 2-bromobenzene O 2-bromophenolarrow_forwardThe following molecule is a :ÓH 2° alcohol 1° amine 1° alcohol 3° amine 3° alcohol 2° aminearrow_forward
- When the conjugate acid of aniline, C6H5NH3+, reacts with the acetate ion, the following reaction takes place: C6H5NH3+(aq)+CH3COO(aq)C6H5NH2(aq)+CH3COOH(aq) If Kafor C6H5NH3+ is 1.35105 and Kafor CH3COOH is 1.86105 , what is K for the reaction?arrow_forward18-18 Propanoic acid and methyl acetate are constitutional isomers, and both are liquids at room temperature. One of these compounds has a boiling point of 141°C; the other has a boiling point of 57°C. Which compound has which boiling point? Explain.arrow_forwardIn terms of polarity, which carbonyl group atom possesses a a. partial positive charge b. partial negative chargearrow_forward
- What products are formed when benzoic acid (C 6H 5COOH) is treated with each base: (a) NaOH; (b) Na 2CO 3; (c) NaHCO 3?arrow_forwardThe following amines have the same molecular formula (C5H13N), but their boiling points are significantly different. Explain why. H `NH2 2-Methylbutan-1-amine Boiling point = 97 °C N-Methylbutan-2-amine Boiling point = 84 °C N-Ethyl-N-methylethan-1-amine Boiling point = 65 °Carrow_forwardList the products of each amine reaction. H CH, —N—CH—сH, + CH, CH, CH;-CH-CH;-C-OHarrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





