
(a)
Interpretation:
An acceptable name should be determined for the following acid:
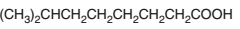
Concept introduction:
There are two acceptable names for a compound.
- IUPAC name
- Common name
Common names use older names of the compounds other than the prefix for carbon skeleton. Most of these names are derived by natural sources of specific compounds.
(b)
Interpretation:
An acceptable name should be determined for the following acid:
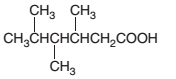
Concept introduction:
There are two acceptable names for a compound.
- IUPAC name
- Common name
Common names use older names of the compounds other than the prefix for carbon skeleton. Most of these names are derived by natural sources of specific compounds.
(c)
Interpretation:
An acceptable name should be determined for the following acid:
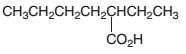
Concept introduction:
There are two acceptable names for a compound.
- IUPAC name
- Common name
Common names use older names of the compounds other than the prefix for carbon skeleton. Most of these names are derived by natural sources of specific compounds.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- butanoic acid+ Ethanolarrow_forwardWhy is it safe for us to consume foods like vinegar that contain acetic acids?arrow_forwardGive the name of a carboxylic acid or carboxylate salt used in each of the following ways: a.As a soap b.As a general food preservative used to pickle vegetables c.As a preservative used in soft drinks d.As a treatment of athletes foot e.As a mold inhibitor used in bread. f.As a food additive noted for its pH buffering abilityarrow_forward
- Name each compound as a phenol.arrow_forwardhelp with carboxylic acid reactionsarrow_forwardSelect the correct systematic name of each ester. H3 C- methyl acetate methyl ethanoate ethyl methanoate 2-propanoate H3C-CH2-CH₂- 3-heptanoate propyl butanoate butyl propanoate propyl butanate -CH3 -CH₂-CH2-CH3arrow_forward
- Determine whether each phrase describes carboxylic acids or esters. Usually have a sour odor Carboxylic acids Do not form hydrogen bonds amongst themselves and have higher vapor pressure Esters Notable for their pleasant fragrances Esters Their reactions with base are known as saponifications Esters Form hydrogen bonds amongst themselves and have lower vapor pressure Carboxylic acids Their reactions with base are known as neutralizations Carboxylic acidsarrow_forwardDefine carboxylationarrow_forwardDraw a structure for cycloheptanecarboxylic acidarrow_forward
- What is/are true regarding the following compound? * COOH IUPAC name is 4-Carboxy-2- pentylbutanoate It is a carboxylic acid It is a carboxy ester IUPAC name is 4-Butanoloxy-2- methylpentanoic acidarrow_forwardWhat is the IUPAC name for the compound shown? OH O 1-ethylcyclohexane-3-carboxylic acid O (2-ethyl)-1-cyclohexylmethanecarboxylic acid O 1-(2-ethyl)-cyclohexylmethanecarboxylic acid O 3-ethylcyclohexanoic acid O 3-ethylcyclohexanecarboxylic acidarrow_forwardAcid Anhydride Amine Thiol Peroxide Nitrile Arenearrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,



