
Concept explainers
(a)
Interpretation:
Acceptable name for the following compound should be determined:
HCONH2
Concept Introduction:
All three carboxylic acids, esters and amides contain carbonyl group (carbon atom connects to oxygen atom via double bond).
In carboxylic acids, there has a carboxylic group (COOH).
In esters, there has an alkoxy group (OR') bonded to the carbonyl carbon.
In amides, there has a nitrogen atom bonded to the carbonyl carbon.
(b)
Interpretation:
Acceptable name for the following compound should be determined:
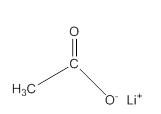
Concept Introduction:
All three carboxylic acids, esters and amides contain carbonyl group (carbon atom connects to oxygen atom via double bond).
In carboxylic acids, there has a carboxylic group (COOH).
In esters, there has an alkoxy group (OR') bonded to the carbonyl carbon.
In amides, there has a nitrogen atom bonded to the carbonyl carbon.
(c)
Interpretation:
Acceptable name for the following compound should be determined:
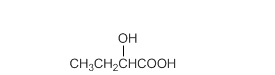
Concept Introduction:
All three carboxylic acids, esters and amides contain carbonyl group (carbon atom connects to oxygen atom via double bond).
In carboxylic acids, there has a carboxylic group (COOH).
In esters, there has an alkoxy group (OR') bonded to the carbonyl carbon.
In amides, there has a nitrogen atom bonded to the carbonyl carbon.
(d)
Interpretation:
Acceptable name for the following compound should be determined:
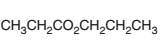
Concept Introduction:
All three carboxylic acids, esters and amides contain carbonyl group (carbon atom connects to oxygen atom via double bond).
In carboxylic acids, there has a carboxylic group (COOH).
In esters, there has an alkoxy group (OR') bonded to the carbonyl carbon.
In amides, there has a nitrogen atom bonded to the carbonyl carbon.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- HCI CH3OH CH₁₂arrow_forwardArrange these compounds in order of increasing boiling point (values in C are 42, 24, 78, and 118). (a) CH3CH2OH (b) CH3OCH3 (c) CH3CH2CH3 (d) CH3COOHarrow_forwardThe common name for the compound CH3-CH2-CH2-O-CH₂-CH3 is ethyl propyl ether O 3-ether pentane 3-pentanol O 3-hexanol O ethyl propyl ketonearrow_forward
- Give the IUPAC name for each structure. Part 1 of 3 CH3(CH2)2CO₂ (CH2)CH3 Part 2 of 3 Part 3 of 3 -CO,—CH,—CH,arrow_forwardGive systematic (IUPAC) name for each compound. H₂C-Cl IUPAC name: CH3 HỌC–CH–CH,-CH-CH,CH, B,C-CH-CH Cl IUPAC name: CHIC IUPAC name:arrow_forwardName the following alcohol. O2-Bromo-4-ethylcyclopentanol 4-Ethyl-2-bromocyclopentanol O1-Ethyl-3-bromo-4-cyclopentanol O1-Bromo-4-ethyl-2-cyclopentanolarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


