
Concept explainers
(a)
Interpretation:
The products formed when the given ester is treated with H2O and
Concept Introduction:
Alcohols are the organic compounds with the general chemical formula of R-OH whereas carboxylic acids are the organic molecules with R-COOH as a general chemical formula.
(b)
Interpretation:
The products formed when the given ester is treated with H2O and
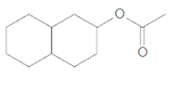
Concept Introduction:
Functional groups are the groups of atoms or atoms which are bonded with the parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester or amide.
Alcohols are the organic compounds with the general chemical formula of R-OH whereas carboxylic acids are the organic molecules with R-COOH as a general chemical formula.
(c)
Interpretation:
The products formed when the given ester is treated with H2O and
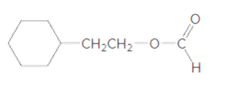
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with the preparation, reactions, and properties of organic compounds. The molecular formula of an organic compound represents the number of bonded atoms with their atomic symbols.
Functional groups are the groups of atoms or atoms which are bonded with the parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester or amide.
Alcohols are the organic compounds with the general chemical formula of R-OH whereas carboxylic acids are the organic molecules with R-COOH as a general chemical formula.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- Give the name of a carboxylic acid or carboxylate salt used in each of the following ways: a.As a soap b.As a general food preservative used to pickle vegetables c.As a preservative used in soft drinks d.As a treatment of athletes foot e.As a mold inhibitor used in bread. f.As a food additive noted for its pH buffering abilityarrow_forwardWhy is it safe for us to consume foods like vinegar that contain acetic acids?arrow_forwardWhat products are formed when each ester is hydrolyzed with water and H 2SO 4?arrow_forward
- Saponification product of butylpropanoate is: Butanoic acid and Propanoic acid B Propanol and Sodium butanoate Butanol and Propanoic acid D Butanol and sodium propanoatearrow_forwardEtoposide, sold as a phosphate derivative with the trade name ofEtopophos, is used for the treatment of lung cancer, testicular cancer,and lymphomas. What products are formed when all of the acetals are hydrolyzed with aqueous acid?arrow_forwardWhat product is formed when each compound is treated with either LiAlH4 (followed by H2O), or NaBH4 in CH3OH?arrow_forward
- Amino acids such as glycine are the building blocks of large molecules called proteins that give structure to muscle, tendon, hair, and nails. What product is formed when glycine is treated with concentrated HCl? What product is formed when glycine is treated with NaOH?arrow_forwardWhat carbonyl compound and amine are formed by the hydrolysis of each compound?arrow_forwardFenofibrate is a cholesterol-lowering medication that is converted to fenofibric acid, the active drug, by hydrolysis during metabolism. What is the structure of fenofibric acid?arrow_forward
- What product is formed when each compound is treated with Tollens reagent (Ag 2O, NH 4OH)?arrow_forward8 What ester is formed when each carboxylic acid is treated with 2-propanol [(CH 3) 2CHOH] in the presence of H 2SO 4?arrow_forwardWhat product is formed when each compound is treated with either Ag2O, NH4OH or Na2Cr2O7, H2SO4, H2O?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

