
(a)
Interpretation:
The products which are formed when the following amide is hydrolyzed with water and NaOH should be determined:
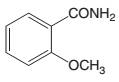
Concept introduction:
Amide has a carbonyl carbon which is linked by a single bond to a nitrogen atom. Amides are
(b)
Interpretation:
The products which are formed when the following amide is hydrolyzed with water and NaOH should be determined:
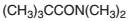
Concept introduction:
Amide has a carbonyl carbon which is linked by a single bond to a nitrogen atom. Amides are amine derivatives of carboxylic acids. Hydrolysis of amide can occur in both acidic and basic media..
(c)
Interpretation:
The products which are formed when following amide is hydrolyzed with water and NaOH should be determined:
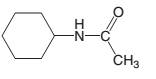
Concept introduction:
Amide has a carbonyl carbon which is linked by a single bond to a nitrogen atom. Amides are amine derivatives of carboxylic acids. Hydrolysis of amide can be occurred in both acidic and basic media..
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- The presence of amides in living organisms is beneficial due its stability which results from being the least reactive carboxylic acid derivative. True or Falsearrow_forwardWhat products are formed when each amide is hydrolyzed with water and HCl?arrow_forwardOxytocin, sold under the trade name Pitocin, is a naturally occurring hormone used to stimulate uterine contractions and induce labor. Classify each amide in oxytocin as 1°, 2°, or 3°. NH2 H2N NH HN 'N H2N NH NH2 S oxytocin но IZarrow_forward
- Classify the amides in dolastatin, an anticancer compound isolated from the Indian seahare Dolabella auricularia, as 1 °, 2°, or 3°.arrow_forwardOxytocin, sold under the trade name Pitocin, is a naturally occurring hormone used to stimulate uterine contractions and induce labor. Classify each amide in oxytocin as 1°, 2°, or 3°.arrow_forwardCompare N-methylacetamide with its amide isomers propanamide and N,N-dimethylformamide. Which do you predict has the highest boiling point? The lowest?arrow_forward
- Give the name of a carboxylic acid or carboxylate salt used in each of the following ways: a.As a soap b.As a general food preservative used to pickle vegetables c.As a preservative used in soft drinks d.As a treatment of athletes foot e.As a mold inhibitor used in bread. f.As a food additive noted for its pH buffering abilityarrow_forwardDraw the structural formula for each amides. - N-Cycloclohexylacetamide - Benzamidearrow_forwardWhat ammonium salt is formed when each amine is treated with HCl? Draw the structure of the resulting salt.arrow_forward
- Give the IUPAC name for the following acid amide: 0 CH3CH2CH(CH3)CH2CH(CH3)CH2C NH2arrow_forwardClassify the specified amides as 1º, 2º, or 3º. Part 1 Select the single best answer. Part 2 Oxytocin, sold under the trade name Pitocin, is a naturally occurring hormone used to stimulate uterine contractions and induce labor. Classify the specified amides in oxytocin as 1º, 2º, or 3º.arrow_forwardWhich of the following contains an amide? HO A. N. B. НО. ОН с. -OH H₂N D.arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning




