
Concept explainers
(a)
Interpretation:
The amide formed when propylamine (CH3CH2CH2NH2) is heated with CH3CH2COOH needs to be determined.
Concept Introduction:
When
General reaction is represented as follows:

(b)
Interpretation:
The amide formed, when propylamine (CH3CH2CH2NH2) is heated with the following carboxylic acid needs to be determined.
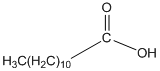
Concept Introduction:
When carboxylic acid is heated with an amine, amide formation can be observed.
General reaction is represented as follows:

(c)
Interpretation:
The amide formed, when propylamine (CH3CH2CH2NH2) is heated with the following carboxylic acid needs to be determined.
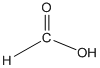
Concept Introduction:
When carboxylic acid is heated with an amine, amide formation can be observed.
General reaction is represented as follows:

(d)
Interpretation:
The amide formed, when propylamine (CH3CH2CH2NH2) is heated with the following carboxylic acid needs to be determined.
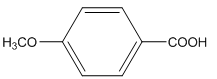
Concept Introduction:
When carboxylic acid is heated with an amine, amide formation can be observed.
General reaction is represented as follows:

Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- An amidification reaction is the reaction of a carboxylic acid with an amine (or ammonia) to produce an amide. Draw the structure of the carboxylic acid from which the following amide could be formed.arrow_forwardName this amine. CH2-CH2-CH-CH2-CH3 NH₂arrow_forwardWhat functional group is NOT present in aspartame, a common artificial sweetener? H₂N `N O Carboxylic acid O None of these O Amine O Ester O Alcohol H COOH LOCH3arrow_forward
- Valacyclovir is an antiviral drug that slows the growth and spread of the herpes virus to help the body fight the infection. Shown below is the structure of the drug. Which of the following functional groups can be found in the structure of valacyclovir? Select one or more: Carboxylic Acid Alcohol Amide Benzene ооооооо Ester Alkyne Amine O Ether Hist HN H₂N NH₂arrow_forward22. How will you name this compound: 3HC-CH2-CH2-CH2-CH2-CH2- NH2? O hexanol amide O hexyl amide O hexyl aminearrow_forwardWhen amines are reacted with bases, they form ammonium salts. Is that true or false?arrow_forward
- H₂C Carboxylic Acid Acid anhydride CHÍNH, CH₂ H₂O H3C Amide CHÍNH, LIAIH, CHÍNH, Acid chloride Amine Esterarrow_forwardPutrescine is produced during the decay and protein breakdown of meats and is responsible for some of the odor found in them. Identify the functional group circled. „NH2 H2N° aldehyde O nitrile amine amide O ketonearrow_forwardDraw the line structures for the carboxylic acid and alcohol that can be used to prepare the ester CH;CH,CO,CH3. Carboxylic acid: Alcohol:arrow_forward
- Alcohols contain which functional group? amine thiol amide hydroxylarrow_forwardTo make an amide, you could react an amine with a(n) alkene ether alcohol acid anhydridearrow_forwardAcetominophen is a widely used and an effective pain reliever. Identify the functional group circled. N. HO aldehyde amide alcohol amine O carbonylarrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





