
Concept explainers
(a)
Interpretation:
The products which are formed when following ester is hydrolyzed with water and H2SO4:
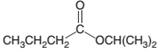
Concept introduction:
Hydrolysis of an ester in an acidic medium releases
(b)
Interpretation:
The products which are formed when the following ester is hydrolyzed with water and H2SO4:
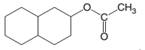
Concept introduction:
Hydrolysis of an ester in an acidic medium releases carboxylic acid and alcohol, whereas hydrolysis of an ester in a basic medium releases carboxylate salt and alcohol. Fisher esterification is the method of ester formation in the presence of acid. Here, all steps are reversible. Therefore, ester hydrolysis in acidic medium is the reversible reaction of Fisher esterification.
(c)
Interpretation:
The products which are formed when following ester is hydrolyzed with water and H2SO4:
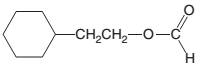
Concept introduction:
Hydrolysis of an ester in an acidic medium releases carboxylic acid and alcohol, whereas hydrolysis of an ester in a basic medium releases carboxylate salt and alcohol. Fisher esterification is the method of ester formation in the presence of acid. Here, all steps are reversible. Therefore, ester hydrolysis in acidic medium is the reversible reaction of Fisher esterification.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- What is the IUPAC name for CH3CH2CH2CH2OHCH3CH2CH2CH2OH? 5-pentanal pentanal 3-butanol 1-butanol 5-pentanol 1-pentanolarrow_forwardDraw the products formed when each ester is hydrolyzed with water and sulfuric acid.arrow_forwardWhat happens to phenol when: a) it reacts with NaOH? b) it reacts with sodium bicarbonate? c) it reacts with Ferric chloride? d) it reacts with Tollen's reagent? e) it undergoes esterification?arrow_forward
- What is the IUPAC name for the following compound? CI 4-ethyl-3,5-dimethylhexanoyl chloride O 3-ethyl-2,4-dimethylhexanoyl chloride 4-ethyl-3,5-dimethylpentanoyl chloride O 3-ethyl-2,4-dimethylpentanoyl chloridearrow_forwardWhat is the correct IUPAC name of the given structure? Br OH CH3 CH3 What is the name of the compound produced by the reaction of benzoic acid with potassium hydroxide? What is the IUPAC name of the ester formed by the reaction of CH3COOH and CH3CH2CH2OH? Which of these products would be formed in the following reaction? H2O NaOH O 1-propanol and butanoic acid O 1-propanol and sodium butanoate O 1-butanol and propanoic acid 1-butanol and sodium propanoatearrow_forwardWhat is the name of this compound? O Phenoxy-2-ethylbutanoic acid O2-ethylbutyl benzoate O benzyl 2-ethylbutanoate Phenyl 2-isopropylbutanoate O Phenyl 2-ethylbutanoatearrow_forward
- Benzaldehyde is the compound principally responsible for the odor of almonds. What product is formed when benzaldehyde is treated with H2, Pd?arrow_forwardWhat is the IUPAC name of the following Organic Compound? OH Ⓒ6-hydroxy-3-propylheptan-2-onc 5-cthanoyloctan-2-ol 3-propylheptanoic acid 03-buytl-3-hydroxyhexan-2-onc 2-hydroxy-5-propylheptan-6-one 9-hydroxydecan-2-oncarrow_forwardWhat is the name of the following carboxylic acid? OH 3-propylbutanoic acid 3-methyl-3-propylpropanoic acid 3-methylpentanoic acid 3-methylhexanoic acidarrow_forward
- What products are formed when aldehydes are oxidized?arrow_forwardWhat is the IUPAC name of the following compound? Ph HO, O 5,5-dimethyl-5-phenylpentanoic acid O 5-methyl-5-phenylhexanoic acid O 2,2-dimethylphenylpropanoic acid O 5,5-dimethyl-5-phenylbutanalarrow_forwardWhat is the correct structure for phenyl benzoate? O PhCO2Ph OCH3CO Ph O PhCH2CO2Ph O PhCH2CO2CH2Pharrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning



