
Concept explainers
Interpretation:
The dicarboxylic acid and
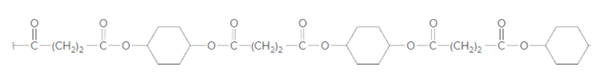
Concept Introduction:
On the basis of type of polymers, polymerization reaction can be two types; addition polymerization and condensation polymerization. The addition polymerization occurs between monomer units with unsaturated bonds between carbon atoms. For example; polymerization of
Condensation polymerization occurs with those monomers which have some functional groups like −OH, -COOH, -NH2 etc. These polymerizations occur with elimination of small molecules such as H2O, CO, and CO2 etc.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
- when ethanoic acid reacts with diethylamine, provide the IUPAC of the major product formedarrow_forwardState the alcohol and carboxylic acid required to form octyl butanoate:arrow_forwardHow many esters can be formed by ethylene glycol with formic and acetic acids? (One or both of the –OH groups of ethylene glycol can react.)arrow_forward
- Draw the structure if the following compounds. benzoic propanoic anhyydride octyl butanoate 30methylhexanoyl chloridearrow_forwardWhat is the general name of the organic species that forms when a carboxylic acis is reacted with a strong base?arrow_forwardwhat happens when benzene diazonium chloride is heated with water?arrow_forward
- What is the product of pentene and water under acidic conditions? Group of answer choices pentanol 2-pentanone pentanoic acid pentanalarrow_forwardIn preparation of methyl orange, what products are prepared?arrow_forward19 What is the name of this molecule? H. heptanal heptanol heptanone heptanoic acidarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





