
(a)
Interpretation:
The acceptable name of following amide should be determined:
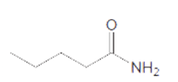
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with the preparation, reactions, and properties of organic compounds are said to be
(b)
Interpretation:
The acceptable name of following amide should be determined:
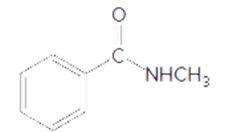
Concept Introduction:
Organic compounds are the compounds that are mainly composed of C and H atoms. The branch of chemistry that deals with the preparation, reactions, and properties of organic compounds are said to be organic chemistry. The molecular formula of the organic compound represents the number of bonded atoms with their atomic symbols.
Functional groups are the groups of atoms or atoms which are bonded with the parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
(c)
Interpretation:
The acceptable name of following amide should be determined:
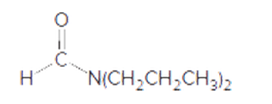
Concept Introduction:
Organic compounds are the compounds which are mainly composed C and H atoms. The branch of chemistry that deals with preparation, reactions, and properties of organic compounds is said to be organic chemistry. The molecular formula of organic compound represents the number of bonded atoms with their atomic symbols.
The structural formula represents all the bonded atoms with
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
- 1. What are the three kinds of alkaloids? Describe each. 2. Classify the following alkaloid according to ring system, draw the structure, then give the natural occurrence. a. betanidine b. peganine C. cocaine d. ergothioneinearrow_forwardexplain the solubility behavior of each representatives amine in water a. aniline b. diethylamine c. N,N-diethylanilinearrow_forwardWhat are the functional groups present in this antibacterial antibiotic? A. Nitro, phenyl, amine, carbonyl, hydroxyl B. Nitro, phenyl, amine, carbonyl, C. Nitro, phenyl, amine, hydroxyl D. Nitro, phenol, amine, carbonyl, hydroxyl A brief explanation would be highly appreciated + upvotearrow_forward
- Draw a structural formula for each amine and amine derivative. Q.) tert-Butylaminearrow_forwardGive an acceptable name for each amine.arrow_forwardExplin how you would separate a mixture containing benzoic acid and a neutral substance, benzophenone. Both compounds are insoluble in water and soluble in ether.arrow_forward
- Synthadotin is a promising anticancer drug in clinical trials. a.Identify the functional groups. b.Classify any amine or amide as 1°, 2°, or 3°. c.At which sites can synthadotin hydrogen bond to another molecule like itself? d. Label two nucleophilic sites. e. Label two electrophilic sites. f. What product is formed when synthadotin is treated with HCl?arrow_forwardDraw a structural formula for each amine and amine derivative. Q.) Benzylaminearrow_forwardWhy free bases of alkaloids are not soluble in polor solvent (water) ?arrow_forward
- Match the description to one of the compounds E– H. a. a compound that contains a 1 ° amine and a 1 ° amide b. a compound that contains a 1 ° amine and a 2 ° amide c. a compound that contains a 2 ° amine and a 3 ° amide d. a compound that contains a 3 ° amine and a 3 ° amidearrow_forwardC. Amides 1. Amidation a. Acetic acid + ammoniaarrow_forwardVI. What ammonium salt is formed when each amine is treated with HCI? Draw the structure of the resulting salt. a. -NH2 b. -CH,NHCH3arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

