
(a)
Interpretation:
The products formed when the following amide is treated with H2O and H2SO4 should be determined:
Concept Introduction:
(b)
Interpretation:
The products formed when the following amide is treated with H2O and H2SO4 should be determined:
Concept Introduction:
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
Amines are the organic compounds with general chemical formula of R-NH2 or R-NH-R whereas carboxylic acids are the organic molecules with R-COOH as general chemical formula.
(c)
Interpretation:
The products formed when the following amide is treated with H2O and H2SO4 should be determined:
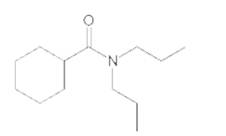
Concept Introduction:
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
Amines are the organic compounds with general chemical formula of R-NH2 or R-NH-R whereas carboxylic acids are the organic molecules with R-COOH as general chemical formula.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Loose Leaf for General, Organic and Biological Chemistry with Connect 2 Year Access Card
- What carbonyl compound and amine are formed by the hydrolysis of each compound?arrow_forwardFenfluramine and phentermine are two components of fen–phen, an appetite suppressant withdrawn from the market in 1997 after it was shown to damage the heart valves in some patients. What products are formed when fenfluramine and phentermine are each treated with acetic acid (CH3CO2H)?arrow_forwardMatch the description to one of the compounds E– H. a. a compound that contains a 1 ° amine and a 1 ° amide b. a compound that contains a 1 ° amine and a 2 ° amide c. a compound that contains a 2 ° amine and a 3 ° amide d. a compound that contains a 3 ° amine and a 3 ° amidearrow_forward
- VI. What ammonium salt is formed when each amine is treated with HCI? Draw the structure of the resulting salt. a. -NH2 b. -CH,NHCH3arrow_forwardWhat ammonium salt is formed when each amine is treated with HCl? Draw the structure of the resulting salt.arrow_forwardErythronolide B is the biological precursor of erythromycin, a broad-spectrum antibiotic. What functional group Erythronolide B does contain? a. b. H₂CH₂C C. H₂C 1 H₂C 2 3 4 O Amide d. Amine OH Erythronolide B Ketone Aldehyde a CH₂ b C d CH₂ OH JCH₂ 'OH OHarrow_forward
- What are the products of the reaction between acetyl chloride and diethylamine? HCl and N,N-diethylacetamide Оа. o b. HCl and diethylamine chloride ос. HCl and N-ethylacetamide d. Diethylacetamide and acidarrow_forward7 8 3: H3C H3C. CH₂ CH3 N,N-diethylpropanamine N-ethyl-pentanamine 3-ethylhexanamine 3-propylpentanaminearrow_forwardList the products of each amine reaction. H b. CH;-N-CH-CH, + HCN CH3arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
