
Concept explainers
(a)
Interpretation:
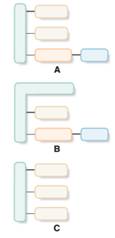
Structures which are aligned with phospholipid should be identified from the given block diagrams.
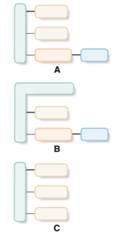
Concept Introduction:
A phospholipid has a phosphorus atom in its lipid structure. Two types of phospholipids are available; phosphoacylglycerols and sphingomyelins.
Phosphoacylglycerols consist of two fatty acids, one phosphate group bonded with low molecular weight alcohol and one glycerol molecule in the structure.
Sphingomyelin derived from a sphingosine molecule that connects to a fatty acid molecule and a phosphate-containing alcohol molecule.
(b)
Interpretation:
Structures that are aligned with triacylglycerol should be identified from the given block diagrams.
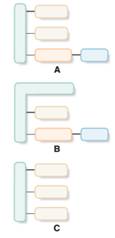
Concept Introduction:
Simple triacylglycerol contains one glycerol molecule and three fatty acid molecules bind with ester bonds.
(c)
Interpretation:
Structures which are aligned with hydrolyzable lipid should be identified from the given block diagrams.
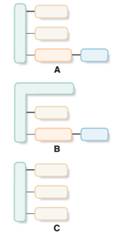
Concept Introduction:
In the presence of acid, base or enzyme, triacylglycerol molecules cleave the ester bonds in it and form fatty acids and glycerol.
(d)
Interpretation:
Structures that are aligned with sphingomyelin should be identified from the given block diagrams.
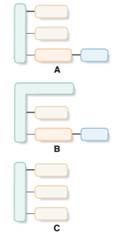
Concept Introduction:
Sphingomyelin derived from a sphingosine molecule that connects to a fatty acid molecule and a phosphate containing alcohol molecule.
(e)
Interpretation:
Structures that are aligned with phosphoacylglycerol should be identified from the given block diagrams.
Concept Introduction:
Phosphoacylglycerols consist of two fatty acids, one phosphate group bonded with a low molecular weight alcohol and one glycerol molecule in the structure.
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
EBK GENERAL, ORGANIC, & BIOLOGICAL CHEM
- 21-100 What are the functions of a cell membrane? To what extent is a bilayer that consists entirely of lipids able to carry out these functions?arrow_forwardFats belong to the class of organic compounds represented by the general formula, RCOOR', where R and R' represent hydrocarbon groups. What is the name of the functional group present in fats? What functional group is common to all saponifiable lipids?arrow_forwardWhat is the basis for deciding if a substance is a lipid?arrow_forward
- Give numerical answers to the following questions about the structure of a cholesterol molecule. a. How many six-membered rings arc present? b. How many amide linkages are present? c. How many hydroxyl substituents are present? d. How many total functional groups are present?arrow_forwardThe following is a block diagram for a glycerophospholipid where the building blocks are labeled with letters and the linkages between building blocks are labeled with numbers. a. Which building blocks are fatty acid residues? b. Which building blocks are alcohol residues? c. Which linkages are ester linkages? d. Which linkages involve a phosphate residue?arrow_forward21-85 Which of the following statements is (are) consistent with what is known about membranes? (a) A membrane consists of a layer of proteins sandwiched between two layers of lipids. (b) The compositions of the inner and outer lipid layers are the same in any individual membrane. (c) Membranes contain glycolipids and glycoproteins. (d) Lipid bilayers are an important component of membranes. (e) Covalent bonding takes place between lipids and proteins in most membranes.arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





