
Concept explainers
Interpretation:
The stereochemistry of the pyruvate reduction shown is to be stated. Whether NADH lose its pro-R or pro-S hydrogen is to be stated. Whether the addition of hydrogen occurs to the Si face or Re face of pyruvate is also to be stated.
Concept introduction:
When an achiral molecule such as
In addition a compound with a sp3 hybridized carbon also is said to be prochiral if by changing one of the attached groups it becomes a chiral centre. If the prochiral centre has two identical atoms, then that atom whose replacement leads to a R chirality centre is said to be pro R and that atom whose replacement leads to a S chirality centre is said to be pro S.
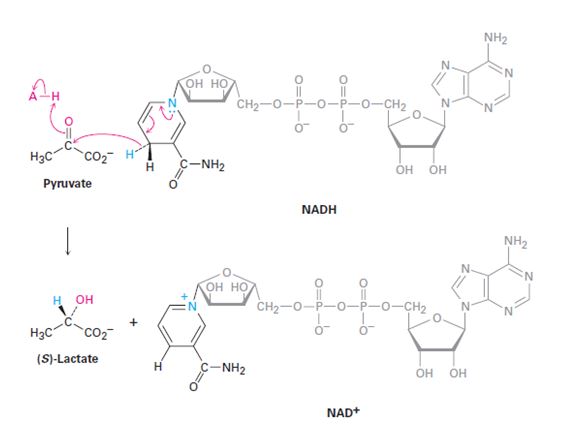
To show:
The stereochemistry of the pyruvate reduction to (s)-lactate shown and to state whether NADH lose its pro-R or pro-S hydrogen and whether the addition of hydrogen occurs to the Si face or Re face of pyruvate.
Trending nowThis is a popular solution!

Chapter 19 Solutions
Organic Chemistry
- In step 7 of fatty-acid biosynthesis (Figure 29-5), dehydration of a -hydroxy thioester occurs to give trans-crotonyl ACP. Is the dehydration a syn elimination or an anti elimination?arrow_forwardAssume that acetyl CoA containing a 14C isotopic label in the carboxyl carbon atom is used as starting material for the biosynthesis of mevalonate, as shown in Figure 27-7. At what positions in mevalonate would the isotopic label appear?arrow_forwardThe aconitase-catalyzed addition of water to cis-aconitate in the citric acid cycle occurs with the following stereochemistry. Does the addition of the OH group occur on the Re or Si face of the substrate? What about the addition of the H? Do the H and OH groups add from the same side of the double bond or from opposite sides?arrow_forward
- Acid-catalyzed dehydration reactions are normally E1 reactions. Why is the acid-catalyzed dehydration in the second reaction of the citric acid cycle an E2 reaction?arrow_forwardIn glycerol metabolism, the oxidation of sn-glycerol 3-phosphate to give dihydroxyacetone phosphate is catalyzed by sn-glycerol-3-phosphate dehydrogenase, with NAD+ as cofactor. The reaction is stereospecific, occurring exclusively on the Re face of the nicotinamide ring.arrow_forwardWhich compound in each of the following pairs is the better reducing agent? a. NADH/H2O b. UQH2/FADH2 c. Cyt c (reduced)/cyt b (reduced) d. FADH2/NADH e. NADH/FMNH2arrow_forward
- draw the chemical structures of the reactant and product of the following reactions: a.) addition of a phosphate group to (ex: phosphorylation of) glycose b.) addition of a phosphate group to (ex: phosphorylation of) fructose-6-phosphate c.) addition of a phosphate group to (ex: phosphorylation of) phosphoenolpyruvatearrow_forwardLinoleic acid is shown below. What makes this fatty acid particularly susceptible to autoxidation? 1. The red CH bond has a low bond dissociation energy because it is doubly allylic. 2. The red CH bond has a high bond dissociation energy because it is doubly allylic. 3. The red CH bond is the most accessible to reaction with O2 because it is the least sterically crowded CH bond. 4. Both 2 and 3.arrow_forwardThe vitamin Niacin is used to form nicotinamide adenosine dinucleotide, which readily shuttles between its oxidized (NAD+) and reduced (NADH) forms. The latter serves as a cellular equivalent to NaBH4. The essential portions of the structures are shown below. Outline a mechanism for the cellular conversion of pyruvate to lactate. (Note: like NaBH4, NADH cannot reduce carboxylic acid carbonyls).arrow_forward
- 17. .Draw the dash-wedge structure for the beta-furanose anomer of the L-ketohexose shown below.arrow_forward1 Provide a mechanism for the following: a. glyceraldehyde-3-phosphate -> dihydroxyacetone phosphatearrow_forwardplease please help me to answer it >>> i have an exam Human xanthine oxidase catalyzes the oxidation of hypoxanthine to xanthine and can furthercatalyze the oxidation of xanthine to uric acid. For the treatment of hyperuricemia and gout,several medications are used to inhibit the activity of xanthine oxidase and reduce theproduction of uric acid. You are a biochemist and just discovered a chemical that can inhibitthe activity of the human xanthine oxidase. When analyzing its mode of inhibition, you foundthat the enzyme inhibitor complex requires 450 J.mol-1 to dissociate and that it displayskinetics somehow similar to noncompetitive inhibition. You sent your inhibitor to the ministryof health for approval as a medication for gout. Based on the data provided, are they going toauthorize it as a medication or not? Explain?arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

