
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19.8D, Problem 19.10P
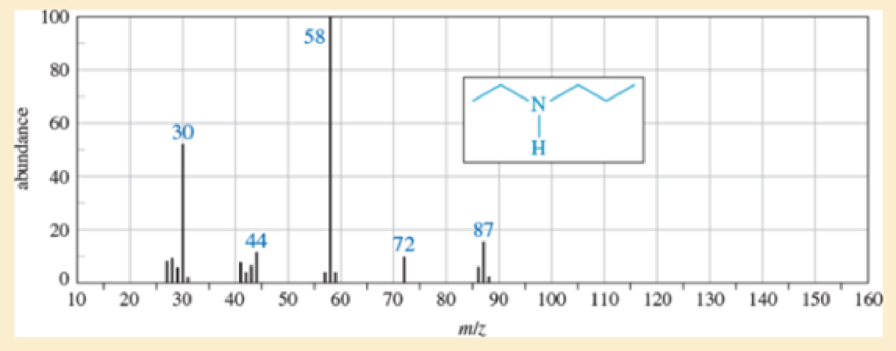
- a. Show how fragmentation occurs to give the base peak at m/z 58 in the mass spectrum of ethyl propyl
amine (N-ethylpropan-1-amine), shown below. - b. Show how a similar cleavage in the ethyl group gives an ion of m/z 72.
- c. Explain why the peak at m/z 72 is much weaker than the one at m/z 58.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Although 2,2-dimethylhexane has a molecular weight of 114, no peak is observed at m/z = 114. The base peak in the mass spectrum occurs at M-57
(a) Draw the fragmentation responsible for the formation of M-57 ion
(b) Explain why this cation is the most abundant ion to pass through the spectrometer.
(c) Explain why no molecular ion survive long enough to be detected.
(d) Can you offer an explanation as to why M-15 peak is not the base peak?
On each mass spectrum above, indicate the base peak and the molecular ion peak (or at least where you would expect to find it) and if the base peak on the mass spectrum for linalool has m/z 71, propose a structure for this fragment ion.
The base peak in the mass spectrum of hexanamide appears at m/z = 59. Draw the ion that corresponds to this mass peak, and show how it is produced from the molecular ion.
Chapter 19 Solutions
Organic Chemistry (9th Edition)
Ch. 19.2A - Prob. 19.1PCh. 19.2B - Prob. 19.2PCh. 19.2B - Give correct names for the following amines:Ch. 19.3 - Prob. 19.4PCh. 19.4 - Prob. 19.5PCh. 19.6 - Rank each set of compounds in order of increasing...Ch. 19.8A - Prob. 19.7PCh. 19.8C - Prob. 19.8PCh. 19.8C - Prob. 19.9PCh. 19.8D - a. Show how fragmentation occurs to give the base...
Ch. 19.10B - Propose a mechanism for nitration of pyridine at...Ch. 19.10B - Prob. 19.12PCh. 19.10C - Prob. 19.13PCh. 19.10C - Prob. 19.14PCh. 19.11 - Propose a mechanism to show the individual...Ch. 19.11 - Prob. 19.16PCh. 19.12 - Give the products expected from the following...Ch. 19.13 - Prob. 19.18PCh. 19.13 - Prob. 19.19PCh. 19.14 - Prob. 19.20PCh. 19.15 - Prob. 19.21PCh. 19.15 - Prob. 19.22PCh. 19.16 - Prob. 19.23PCh. 19.17 - Prob. 19.24PCh. 19.17 - Prob. 19.25PCh. 19.18 - Prob. 19.26PCh. 19.19 - Prob. 19.27PCh. 19.20A - Addition of one equivalent of ammonia to...Ch. 19.20A - Prob. 19.29PCh. 19.20B - Show how you would accomplish the following...Ch. 19.20C - Prob. 19.31PCh. 19 - For each compound, 1. classify the...Ch. 19 - Prob. 19.33SPCh. 19 - Within each structure, rank the indicated...Ch. 19 - In each pair of compounds, select the stronger...Ch. 19 - Which of the following compounds are capable of...Ch. 19 - Complete the following proposed acid-base...Ch. 19 - Predict the products of the following reactions:...Ch. 19 - Prob. 19.39SPCh. 19 - Show how m-toluidine can be converted to the...Ch. 19 - The mass spectrum of tert-butylamine follows shows...Ch. 19 - Prob. 19.42SPCh. 19 - The following drugs are synthesized using the...Ch. 19 - Prob. 19.44SPCh. 19 - Synthesize from benzene. (Hint: All of these...Ch. 19 - Propose mechanisms for the following reactions.Ch. 19 - Prob. 19.47SPCh. 19 - Prob. 19.48SPCh. 19 - Prob. 19.49SPCh. 19 - Show how you can synthesize the following...Ch. 19 - Prob. 19.51SPCh. 19 - The alkaloid coniine has been isolated from...Ch. 19 - A chemist is summoned to an abandoned...Ch. 19 - Pyrrole undergoes electrophilic aromatic...Ch. 19 - Prob. 19.55SPCh. 19 - Prob. 19.56SPCh. 19 - An unknown compound shows a weak molecular ion at...Ch. 19 - A compound of formula C11H16N2 gives the IR,...Ch. 19 - (A true story.) A drug user responded to an ad...Ch. 19 - Prob. 19.60SPCh. 19 - Prob. 19.61SPCh. 19 - Prob. 19.62SPCh. 19 - Prob. 19.63SPCh. 19 - Prob. 19.64SPCh. 19 - Prob. 19.65SP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The base peak in the mass spectrum of benzyl bromide (PhCH2Br) has a m/z of 91. Draw the structure of this fragment ion.arrow_forward23. Consider the mass spectrum of 2-bromopentane, which exhibits a molecular ion peak at m/z = 150 (a) One fragment appears at M−79. Would you expect a signal at M−77 that is equal in height to the M−79 peak? Explain. Draw the structure of this fragment.arrow_forwardDraw the mass spectrum fragment that observed at m/z 91arrow_forward
- 2,3-Dimethylbutane and 2,2-dimethylbutane have the same molecular ion in the mass spectrum, but only one of these isomers gives a significant fragment at m/z = 57. (a) Which isomer shows an intense peak at m/z = 57? (b) Propose a structure for the ion that gives rise to this peak. (c) The base peak in the mass spectrum of the other isomer occurs at m/z = 43. What ion gives rise to this peak?arrow_forwardIdentify the expected base peak in the mass spectrum of 2,2,3-trimethylbutane. Draw the fragment associated with this peak and explain why the base peak results from this fragment.arrow_forwardAnswer the following questions using the mass spectrum and peak intensity data shown below. (a) What is the m/z of the M+ peak? (b) What is the m/z of the base peak? (c) Estimate the number of carbons in the compound. Show your work! (d) Does the molecule contain a N, Cl or Br? Explain. (e) Which of the compounds below could produce the given spectrum? Explain. (f) Draw the ion that corresponds to the base peak, and show how it is produced from the molecular ion.arrow_forward
- the electron ionization mass spectrum of a small neutral molecule shows a base peak at 27 and a smaller peak at 26 with a relative abundance of 20%. Deduce the structure of the unknown compound. Step 3: if the molecular ion is odd or even, this indicates the number of nitrogens. The mass spec data shows an ____ (odd or even) molecualr weight.arrow_forwardFrom the given IR and mass spectra of the unknown compound: 1. Which element is surely present in the compound? a. F b. O c. Br d. N 2. Which statement is true? a. It has no saturation b. It is an acid anhydride c. It contains -CH2- next to benzene ring d. All are falsearrow_forwardA peak appears at m/z = 83 in the mass spectrum of hept-3-ene. Show how the fragmentation of hept-3-ene’s molecular ion produces the ion that gives rise to this peak. Why do you think the peak at m/z = 83 is smaller than the one at m/z = 69?arrow_forward
- Draw all four resonance forms of the fragment at m>z 73 in the mass spectrum of pentanoic acidarrow_forwardFrom the mass spectrum of Octane 1.Which peak represents the molecular ion? Label the peak with letter A at the top. 2. Which peak is the base peak? Label the peak with letter B at the top. 3. What is the name of the fragment responsible for the base peak? 4. Draw the structure of the fragment that produces the base peakarrow_forwardWhat fragment is responsible for the base peak in the MS spectra of 4-phenyl-2-butanone, 1-phenyl-1-butanone, and 1-phenyl-2-butanonearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY