
Organic Chemistry
11th Edition
ISBN: 9781118133576
Author: T. W. Graham Solomons, Craig Fryhle
Publisher: Wiley, John & Sons, Incorporated
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21, Problem 34P
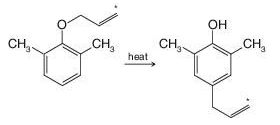
Explain why, in the case shown, the allyl group has migrated with no change having occurred in the position of the labeled carbon atom within the allyl group:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
comparing to this two ketone, which ketone has the most stable π system? Explain on the basis of the carbonyl group.
How do I go by this problem?
Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE.
Give the name of the following compound:
Chapter 21 Solutions
Organic Chemistry
Ch. 21 - PRACTICE PROBLEM
21.1 If we examine Table 21.1, we...Ch. 21 - PRACTICE PROBLEM If we examine Table 21.1, we see...Ch. 21 - Prob. 3PPCh. 21 - PRACTICE PROBLEM
21.4 Predict the products of each...Ch. 21 - Prob. 5PPCh. 21 - PRACTICE PROBLEM
21.6 What are compounds A and B...Ch. 21 - Prob. 7PPCh. 21 - PRACTICE PROBLEM Outline a possible synthesis of...Ch. 21 - PRACTICE PROBLEM 1-Fluoro-2,4-dinitrobenzene is...Ch. 21 - PRACTICE PROBLEM
21.10 When o-chlorotoluene is...
Ch. 21 - PRACTICE PROBLEM When 2-bromo-1,3-dimethylbenzene...Ch. 21 - PRACTICE PROBLEM (a) Outline a step-by-step...Ch. 21 - Rank the following in order of increasing acidity.Ch. 21 - Prob. 14PCh. 21 - Prob. 15PCh. 21 - Describe a simple chemical test that could be used...Ch. 21 - Prob. 17PCh. 21 - Predict the product of the following reactions.Ch. 21 - 21.19 A synthesis of the β-receptor blocker called...Ch. 21 - Prob. 20PCh. 21 - When m-chlorotoluene is treated with sodium amide...Ch. 21 - Prob. 22PCh. 21 - Prob. 23PCh. 21 - Prob. 24PCh. 21 - Prob. 25PCh. 21 - Prob. 26PCh. 21 - Prob. 27PCh. 21 - Prob. 28PCh. 21 - Prob. 29PCh. 21 - Prob. 30PCh. 21 - Prob. 31PCh. 21 - 21.32 A compound X (C10H14O) dissolves in aqueous...Ch. 21 - 21.33 Compound Z (C5H10O) decolorizes bromine in...Ch. 21 - Explain why, in the case shown, the allyl group...Ch. 21 - In protic solvents the naphthoxide ion (I) is...Ch. 21 - Prob. 36PCh. 21 - Prob. 37PCh. 21 - Prob. 38PCh. 21 - Prob. 39PCh. 21 - Prob. 40PCh. 21 - 21.41 Compound W was isolated from a marine...Ch. 21 - 21.42 Phenols generally are not changed on...Ch. 21 - 21.43 Open the molecular model file for benzyne...Ch. 21 - Which of the following would be the strongest...Ch. 21 - What products would you expect from the following...Ch. 21 - Prob. 3QCh. 21 - Prob. 4QCh. 21 - 21.5 Complete the following synthesis:
Ch. 21 - Prob. 6QCh. 21 - 21.7 Select the stronger acid.
Additional Science Textbook Solutions
Find more solutions based on key concepts
Which property of rubbing alcohol is a chemical property? a) its density (0.786 g/cm3) b) its flammability c) i...
Chemistry: A Molecular Approach
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
21.32 Describe the flotation process for concentrating a metal sulfide ore, and explain why this process would ...
Chemistry (7th Edition)
a. Prepare a molecular orbital energy-level diagram for NO showing clearly how the atomic orbitals interact to ...
Inorganic Chemistry
Le Châtelier's Principle and Changes of State
11.67 What do we mean by the position of equilibrium?
Chemistry: The Molecular Nature of Matter
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- How many non-equivalent carbons exist in the following compound? (Give a numberarrow_forwardCarbonyl-containing compounds are equally important in biological molecules. The following line diagram is one example: What is the IUPAC name of the above molecule? Type your answer in, ignoring any isomerism that may exist in this line diagram. You will need to submit a handwritten response to the next part of this question (see below). Label the question number (Q7) and show your answer legibly and clearly. Your handwritten response for this question will be awarded up to Handwritten response Question 7: a. Write a reaction equation between the above compound and propan-1-ol to form a hemiacetal. b. Combine the hemiacetal from part (a) with a second molecule of propan-1-ol and draw the structure of the resulting acetal. In your answer include the structural formulas, preferably as line diagrams, of all organic reactants and products.arrow_forwardCategorize the following molecules as aromatic, anti-aromatic, or non-aromatic by adding the assigned letters to the corresponding category. Assume each molecule is planar.arrow_forward
- Is it logical to conclude that compounds ending in -al are generally aldehyde and those compounds which end in -one can be classified as ketones? Why or why not? Answer in more than 5 sentences.arrow_forward1. The structure of pyridine and pyrrole is given below along with the dipole moment of each molecule. Explain why the nitrogen is the negative end of the dipole in pyridine, while in pyrrole it is the positive end of the dipole. Use structures to support your argument. Pyridine Pyrrolearrow_forwardLabel the following as aromatic, antiaromatic, or nonaromatic.arrow_forward
- Each molecule in this problem can be drawn as a hybrid of five contributing structures: two Kekulé structures and three that involve creation and separation of unlike charges. Draw these five contributing structures for molecule. Q.) Phenolarrow_forwardAlthough we normally think of acetic acid as an acid, it can also act as a weak base. Treated with a strong acid, it can become protonated. The conjugate acid of acetic acid is shown. H₂C :O-H H* Draw curved arrows for resonance structure 1. H₂C Derive a resonance structure for this ion which, taken with the given structure, would show that the two -OH groups are equivalent, the two C-O bonds are equivalent, and the positive charge is shared equally by the two oxygens. с The skeleton for the second resonance structure is provided. Add the bond(s), charge(s), and unshared pairs of electrons. Then draw the curved arrows to show the flow of electrons that transforms each resonance structure into the other. : OH || H3C- I.. -H OH :0 -H Complete the structure of resonance structure 2 and draw curved arrows to transform it to resonance structure 1. *K H₂C -C OH OHarrow_forwardDraw a resonance structure, complete with all formal charges and lone (unshared) electron pairs, that shows the resonance interaction of the cyano with the ortho position in benzonitrile. CEN benzonitrile • You do not have to consider stereochemistry. • Include all valence lone pairs in your answer. • In cases where there is more than one answer, just draw one.arrow_forward
- The carbon–carbon bond lengths in naphthalene are not equal. Use a resonance argument to explain why bond (a) is shorter than bond (b).arrow_forwardWhich of the following is correct about thioethers? They are more reactive than their corresponding ethers. They are partially responsible for “morning breath.” They are organic compounds in which a sulfur is bonded to two carbon atoms by a single bond. All of the abovearrow_forwardWhich of the following resonance structures is the greatest contributor to the resonance hybrid?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY