
Organic Chemistry
11th Edition
ISBN: 9781118133576
Author: T. W. Graham Solomons, Craig Fryhle
Publisher: Wiley, John & Sons, Incorporated
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21, Problem 35P
In protic solvents the naphthoxide ion (I) is alkylated primarily at position 1 (C-alkylation) whereas in
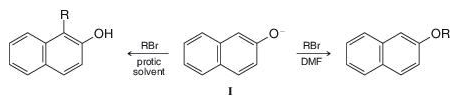
Why does the change in solvent make a difference?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Part III, Reaction Mechanisms
16. (15 pts) Show the mechanism for the following transformation.
осно
aq NaOH
H3O+
Мон
+
H3C-OH
OH
Acyl transfer (nucleophilic substitution at carbonyl) reactions proceed in two stages via a "tetrahedral intermediate." Draw the
tetrahedral intermediate as it is first formed in the following reaction.
H3C
NH₂
HCI/H₂O
reflux
• You do not have to consider stereochemistry.
• Include all valence lone pairs in your answer.
• Do not include counter-ions, e.g., Na+, I, in your answer.
• In cases where there is more than one answer, just draw one.
In the given condensed formula, I understand that there will be a carbocation rearrangement due to excess hydrogen and an E1 mechanism, however, I have trouble in understanding where this bond between the nucleophile and electrophile is formed.
Chapter 21 Solutions
Organic Chemistry
Ch. 21 - PRACTICE PROBLEM
21.1 If we examine Table 21.1, we...Ch. 21 - PRACTICE PROBLEM If we examine Table 21.1, we see...Ch. 21 - Prob. 3PPCh. 21 - PRACTICE PROBLEM
21.4 Predict the products of each...Ch. 21 - Prob. 5PPCh. 21 - PRACTICE PROBLEM
21.6 What are compounds A and B...Ch. 21 - Prob. 7PPCh. 21 - PRACTICE PROBLEM Outline a possible synthesis of...Ch. 21 - PRACTICE PROBLEM 1-Fluoro-2,4-dinitrobenzene is...Ch. 21 - PRACTICE PROBLEM
21.10 When o-chlorotoluene is...
Ch. 21 - PRACTICE PROBLEM When 2-bromo-1,3-dimethylbenzene...Ch. 21 - PRACTICE PROBLEM (a) Outline a step-by-step...Ch. 21 - Rank the following in order of increasing acidity.Ch. 21 - Prob. 14PCh. 21 - Prob. 15PCh. 21 - Describe a simple chemical test that could be used...Ch. 21 - Prob. 17PCh. 21 - Predict the product of the following reactions.Ch. 21 - 21.19 A synthesis of the β-receptor blocker called...Ch. 21 - Prob. 20PCh. 21 - When m-chlorotoluene is treated with sodium amide...Ch. 21 - Prob. 22PCh. 21 - Prob. 23PCh. 21 - Prob. 24PCh. 21 - Prob. 25PCh. 21 - Prob. 26PCh. 21 - Prob. 27PCh. 21 - Prob. 28PCh. 21 - Prob. 29PCh. 21 - Prob. 30PCh. 21 - Prob. 31PCh. 21 - 21.32 A compound X (C10H14O) dissolves in aqueous...Ch. 21 - 21.33 Compound Z (C5H10O) decolorizes bromine in...Ch. 21 - Explain why, in the case shown, the allyl group...Ch. 21 - In protic solvents the naphthoxide ion (I) is...Ch. 21 - Prob. 36PCh. 21 - Prob. 37PCh. 21 - Prob. 38PCh. 21 - Prob. 39PCh. 21 - Prob. 40PCh. 21 - 21.41 Compound W was isolated from a marine...Ch. 21 - 21.42 Phenols generally are not changed on...Ch. 21 - 21.43 Open the molecular model file for benzyne...Ch. 21 - Which of the following would be the strongest...Ch. 21 - What products would you expect from the following...Ch. 21 - Prob. 3QCh. 21 - Prob. 4QCh. 21 - 21.5 Complete the following synthesis:
Ch. 21 - Prob. 6QCh. 21 - 21.7 Select the stronger acid.
Additional Science Textbook Solutions
Find more solutions based on key concepts
Experiments to determine the local convection heat transfer coefficient for uniform flow normal to a heated cir...
Fundamentals of Heat and Mass Transfer
3. Drag out your general chemistry book and solve any four stoichiometry problems dealing with a limiting reage...
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
Q3. Which change is a physical change?
a) Wood burning
b) Iron rusting
c) Dynamite exploding
d) Gasoline ev...
Chemistry: A Molecular Approach (4th Edition)
The following reaction has a value of G = 2.1kJ/mol(0.50kcaI/mol). CH3Br + H2S CH3 SH + HBr a. Calculate Keq a...
Organic Chemistry (9th Edition)
Predict the major product for each of the following reactions:
Organic Chemistry As a Second Language: Second Semester Topics
31. Consider the reaction.
Complete the table.
Rate
-0.012 M/s
Chemistry: Structure and Properties (2nd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the major organic product of the reaction. Indicate the stereochemistry via wedge/dash bonds, including explicit H and D atoms, at the stereogenic center. Omit byproducts such as salts or methanol. Br NaOCH CH OH Harrow_forward1) Draw the complete electron-pushing arrow mechanism for the following reductions. Explain, using resonance contributors (structures), the regiochemistry that results in each case. OMe Na, MeOH ? NH3 CHO Na, MeOH ? NH3arrow_forwardGive detailed Solution with explanation neededarrow_forward
- Liarrow_forwardNucleophilic aromatic substitution involves the formation of a resonance-stabilized carbanion intermediate called a Meisenheimer complex as the nucleophile attacks the ring carbon carrying the eventual leaving group; electron-withdrawing groups ortho and/or para to the site of attack help to stabilize this structure via resonance. For the reaction below, draw the structure of the stabilized reaction intermediate in the box below. CI F3C CF3 NO₂ F3C N CF3 NO₂ • You do not have to consider stereochemistry. • Draw the Meisenheimer complex with a formal charge of 0 on the nucleophilic atom. If more than one resonance structure is possible, only draw the most important one.arrow_forwardMulti-step synthesis. Give the reagents over the arrows and the intermediates between thearrows that are required to synthesize the product compound on the left from the reactant givenon the right for each retrosynthetic analysis.arrow_forward
- ol The conjugate addition of an alkyl group to an a,ß-unsaturated ketone is one of the more useful 1,4-addition reactions, just as direct addition of a Grignard reagent is one of the more useful 1,2-addition reactions. Conjugate addition occurs via the use of a lithium dialkylcopper reagent, also called a Gilman reagent. The mechanism is thought to involve the addition of the dialkylcopper anion to the B- carbon to give a copper-containing intermediate. Transfer of an alkyl group to carbon and release of a neutral alkylcopper species completes the reaction. Draw curved arrows to show the movement of electrons in this step of the mechanism. Arrow-pushing Instructions سلم NOC XT Ph :0: CH₂ 1. (CH3CH₂)2CuLi 2. H₂O* CH₂CH3 Cu CH₂CH3 Ph Cu CH3 CH₂CH3 CH₂CH3 8aarrow_forwardPlease help me with the mechanism with arrows.arrow_forwardОН ARSO3H + H20 Dihydropyran is synthesized by treating tetrahydrofurfuryl alcohol with an arenesulfonic acid, ArSO,H. Draw curved arrows to show the movement of electrons in this step of the reaction mechanism. Arrow-pushing Instructions + OH ОН H.arrow_forward
- Give a reasonable mechanism for this reaction. Explain why it is exothermic. H+ хо Дон H2Oarrow_forward4. A student performed the following sequence of reactions: an oxetane opening, followed by a Williamson ether synthesis; however, the student forgot to write down which alkyl halide they used for the second step of the Williamson ether synthesis, so they do not know what the final product should be. Luckily, a 'H NMR spectrum was obtained for the final product. Determine the product of each step, as well as the alkyl halide that the student added. Draw the mechanism for each step below. MgBr 1. 1. NaH 2. H30* 2. 'H NMR of final product: | = 6 | = 3 | = 2 |= 2 | = 2 3.0 2.5 2.0 1.5 |= 3 | = 2 6 4 2 Mechanism:arrow_forwardDraw the major organic product of the reaction. Indicate the stereochemistry via wedge‑and‑dash bonds, including explicit HH and DD atoms, at the stereogenic center. Omit byproducts such as salts or methanol.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY