
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2.12, Problem 19P
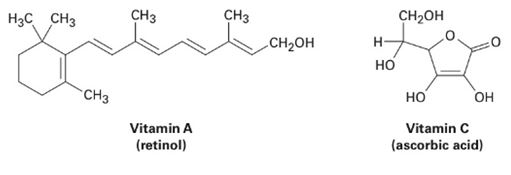
Of the two vitamins A and C, one is hydrophilic and water-soluble while the other is hydrophobic and fat-soluble. Which is which?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
There are two isomeric triacylglycerol molecules whose components are glycerol, one palmitic acid unit, and two stearic acid units. Draw the structures of both, and explain how they differ.
How many constitutional isomers are possible for a triglyceride containing one molecule each of palmitic acid, oleic acid, and stearic acid? (b) Which of these constitutional isomers are chiral?
How many grams of sucrose (C12H22O11) are present in 185 ML of a 2.5 0M sucrose solution?
Chapter 2 Solutions
Organic Chemistry
Ch. 2.1 - Prob. 1PCh. 2.1 - Prob. 2PCh. 2.1 - Use the electronegativity values shown in Figure...Ch. 2.1 - Look at the following electrostatic potential map...Ch. 2.2 - Ethylene glycol, HOCH2CH2OH, may look nonpolar...Ch. 2.2 - Make three-dimensional drawings of the following...Ch. 2.3 - Calculate formal charges for the nonhydrogen atoms...Ch. 2.3 - Organic phosphate groups occur commonly in...Ch. 2.6 - Which of the following pairs of structures...Ch. 2.6 - Draw the indicated number of resonance forms for...
Ch. 2.7 - Nitric acid (HNO3) reacts with ammonia (NH3) to...Ch. 2.8 - Prob. 12PCh. 2.8 - Amide ion, H2N-, is a much stronger base than...Ch. 2.9 - Prob. 14PCh. 2.9 - Prob. 15PCh. 2.9 - Prob. 16PCh. 2.11 - Using curved arrows, show how the species in part...Ch. 2.11 - Prob. 18PCh. 2.12 - Of the two vitamins A and C, one is hydrophilic...Ch. 2.SE - Prob. 20VCCh. 2.SE - The following model is a representation of...Ch. 2.SE - cis-l, 2-Dichloroethylene and trans-1,...Ch. 2.SE - The following molecular models are representations...Ch. 2.SE - Predict the product(s) of the acid/base reactions...Ch. 2.SE - Use curved arrows to draw the protonated form of...Ch. 2.SE - Prob. 26MPCh. 2.SE - Double bonds can also act like Lewis bases,...Ch. 2.SE - Prob. 28APCh. 2.SE - Use the electronegativity table given in Figure...Ch. 2.SE - Which of the following molecules has a dipole...Ch. 2.SE - Prob. 31APCh. 2.SE - Phosgene, C12C=O, has a smaller dipole moment than...Ch. 2.SE - Prob. 33APCh. 2.SE - Methanethiol, CH3SH, has a substantial dipole...Ch. 2.SE - Calculate the formal charges on the atoms shown in...Ch. 2.SE - Assign formal charges to the atoms in each of the...Ch. 2.SE - Which of the following pairs of structures...Ch. 2.SE - Prob. 38APCh. 2.SE - 1, 3-Cyclobutadiene is a rectangular molecule with...Ch. 2.SE - Alcohols can act either as weak acids or as weak...Ch. 2.SE - The O-H hydrogen in acetic acid is more acidic...Ch. 2.SE - Draw electron-dot structures for the following...Ch. 2.SE - Write the products of the following acid-base...Ch. 2.SE - Rank the following substances in order of...Ch. 2.SE - Which, if any, of the substances in Problem 2-44...Ch. 2.SE - The ammonium ion (NH4+, pKa = 9.25) has a lower...Ch. 2.SE - Prob. 47APCh. 2.SE - Prob. 48APCh. 2.SE - Calculate Ka values from the following pka’s:...Ch. 2.SE - Calculate pKa values from the following Ka’s:...Ch. 2.SE - What is the pH of a 0.050 M solution of formic...Ch. 2.SE - Prob. 52APCh. 2.SE - Maleic acid has a dipole moment, but the closely...Ch. 2.SE - Assume that you have two unlabeled bottles, one of...Ch. 2.SE - Identify the acids and bases in the following...Ch. 2.SE - Which of the following pairs represent resonance...Ch. 2.SE - Draw as many resonance structures as you can for...Ch. 2.SE - Carbocations, which contain a trivalent,...Ch. 2.SE - We’ll see in the next chapter that organic...Ch. 2.SE - The azide functional group, which occurs in...Ch. 2.SE - Phenol, C6H5OH, is a stronger acid than methanol,...Ch. 2.SE - Thiamin diphosphate (TPP), a derivative of vitamin...Ch. 2.SE - Determine if each compound or ion below has a...Ch. 2.SE - Prob. 64APCh. 2.SE - Prob. 65APCh. 2.SE - Draw the conjugate base for each compound below...Ch. 2.SE - 1, 1, 1-Trichloroethanol is an acid more than 1000...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A mixture of triacylglycerol molecules contains 67% SFAs, 28% MUFAs, and 5% PUFAs and has a melting point of 32C. a. Should this mixture be classified as a fat or an oil? b. Explain the basis for the answer given in part a.arrow_forwardArrange the following in increasing melting points. Myristic acid, palmitic acid, oleic acid, stearic acid, and linoleic acid. Explain why.arrow_forwardAt the end of 2012, global population was about 7.0 billionpeople. What mass of glucose in kg would be needed toprovide 1500 Cal/person/day of nourishment to the globalpopulation for one year? Assume that glucose is metabolizedentirely to CO21g2 and H2O1l2 according to the followingthermochemical equation:arrow_forward
- Which of the following is not reduced when Benedict’s solution is boiled? Maltose Fructose Sucrose Lactosearrow_forwardWhen a fish is cooked the tissue changes from a soft consistency to a firm one. A similar change takes place when a fish is pickled by soaking it in vinegar/lime juice. In fact some people prefer fish for eating by this later pickling process (Ceviche) instead of cooking. Explain how these two processes give somewhat similar result) Discuss it in terms of protein structure.arrow_forwardAn artificial cell consisting of an aqueous solution enclosed in a selectively permeable membrane (but with no cell wall) is immersed in a beaker containing an aqueous solution. The outside environment concentration consists of 0.01 M glucose and the inside of the cell has a concentration of 5.0 M glucose. The plasma membrane is permeable to water and monosaccharides, but impermeable to the disaccharides. Complete the following for the image below: Is the solution inside the cell isotonic, hypotonic, or hypertonic? Is the glucose going down or against its concentration gradient? Draw solid arrow to indicate the movement of the solute into or out of the cell. Draw a dashed arrow to show the movement of water. Will the artificial cell become lyse, shrivel, or stay the same? the sub-parts to be solved.arrow_forward
- A hibernating bear accumulates 25 kg of a triglyceride (846 g/mol) comprising the following fatty acids: C16, C17 and C18∆9. Calculate how many grams of β-hydroxybutyrate (104 g/mol) the bear can produce from the triglyceride during hibernation? (No other information provided).arrow_forwardBoth triglycerides shown below contained fatty acids that are 16 carbon long. However, one is classified as a fat, and the other is regarded as the oil. (A) How does one distinguish fat from oils? (B) Which triglyceride is the fat? Which one is oil?arrow_forwardHow many constitutional isomers are possible for a triglyceride containing one molecule each of palmitic acid, oleic acid, and stearic acid?arrow_forward
- A bromine test performed on a triacylglyceride and a fatty acid revealed a positive test for one, and a negative test for the other. Describe how this test works, what it is for, and what it says about the difference between the two compounds.arrow_forward(a) Calculate the osmotic pressure in atm. and osmolarity in aqueous solution containing 0.15 mol L-1 KCl, 0.01 mol L-1 of CaCl2 and 0.1 mol L-1 of disaccharide lactose at 20 oC. (b) What will be the osmotic pressure and osmolarity in this solution if all lactose is hydrolysed to monosaccharides glucose and galactose?arrow_forwardFimbriae, and pili are all examples of structures used by microbes for __________. movement adhesion multiplying none of the abovearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY