
Concept explainers
a. Use bond energies (see Table 3-3) to estimate ΔH for the reaction of two molecules of glycine to form a peptide linkage.
b. Would you predict ΔS to favor the formation of peptide linkages between two molecules of glycine?
c. Would you predict the formation of proteins to be a spontaneous process?
(a)
Interpretation: By using given bond energies the enthalpy
Concept introduction: When two or more amino acids are linked in a chain, a compound called peptide is formed in which the carboxyl group of each acid is joined to the amino group of the next forming a
To determine: The
Answer to Problem 130AE
Answer
The
Explanation of Solution
Explanation
The enthalpy
The reaction between two molecules of glycine to form peptide linkage is,
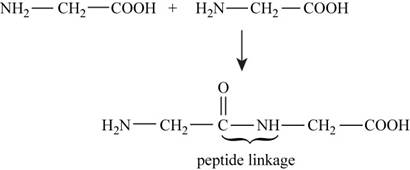
Figure 1
The enthalpy of the reaction is calculated by using the formula,
In the given reaction shown in Figure 1 bonds are broken and formed during peptide formation and energy is absorbed and released. The bond energies are shown in (Table 3-3) respectively.
Therefore, the energy required to break the bonds is calculated as,
Similarly, the energy released when the bonds are formed are calculated as,
Therefore, the total enthalpy for the reaction is calculated by using the formula,
Hence, the total enthalpy of formation for the reaction of two molecules of glycine to form a peptide linkage is positive. It is an endothermic reaction.
(b)
Interpretation: By using given bond energies the enthalpy
Concept introduction: When two or more amino acids are linked in a chain, a compound called peptide is formed in which the carboxyl group of each acid is joined to the amino group of the next forming a
To determine: The entropy
Answer to Problem 130AE
Answer
The entropy
Explanation of Solution
Explanation
The entropy
The reaction between two molecules of glycine to form a peptide linkage is found to be an endothermic process because of the positive value of enthalpy. In the system an endothermic process absorbs heat from the surrounding and therefore, decreases the entropy of the surrounding because the molecular motion in the surrounding decreases. Therefore, for the given reaction
(c)
Interpretation: By using given bond energies the enthalpy
Concept introduction: When two or more amino acids are linked in a chain, a compound called peptide is formed in which the carboxyl group of each acid is joined to the amino group of the next forming a
To determine: The formation of proteins is a spontaneous process or not.
Answer to Problem 130AE
Answer
Formation of proteins is a non-spontaneous process.
Explanation of Solution
Explanation
The formation of proteins is a non-spontaneous process.
The formation of proteins occurs when large numbers of amino acids are joined through peptide linkage. The formation of peptide linkage is a non-spontaneous process as stated above due to the negative value of
Want to see more full solutions like this?
Chapter 22 Solutions
Chemistry
- What functional groups are found in all amino acids? How many different amino acids are found in naturally occurring proteins?arrow_forwardWhy is complete hydrolysis of a protein not also protein denaturation?arrow_forwardDo the amino acid sequences: valine-asparagine and asparagine-valine represent the same compound? Explain.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





