
a)
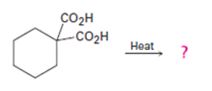
Interpretation:
The products of the reaction shown are to be given.
Concept introduction:
Compounds having two carboxyl groups attached to a carbon readily undergo decarboxylation, when heated, to yield monocarboxylic acids.
To give:
The products of the reaction shown.
b)
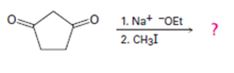
Interpretation:
The product of the reaction shown is to be given.
Concept introduction:
In the first reaction the base abstracts a proton to yield an enolate ion. In the second reaction alkylation takes place.
To give:
The product of the reaction shown.
c)
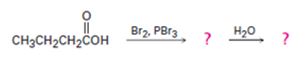
Interpretation:
The products of the reaction shown are to be given.
Concept introduction:
When carboxylic acids are treated with Br2 and PBr3, bromination occurs at the carbon α- to the carboxyl group and the acid group also is converted into an acyl bromide. When treated with water the acyl bromide gets hydrolyzed to yield the free acid.
To give:
The products of the reaction shown.
d)
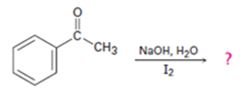
Interpretation:
The products of the reaction shown are to be given.
Concept introduction:
Methyl
To give:
The products of the reaction shown.
Want to see the full answer?
Check out a sample textbook solution
Chapter 22 Solutions
Organic Chemistry
- Predict the product(s) and provide the mechanism for each reaction below.arrow_forward. Predict the products of the following reactions. When more than one product is expected, predict which will be the major product?arrow_forwardPredict the products of the following reactions.(a) cyclohexylmethanol + TsCl/pyridine (b) product of (a) + LiAlH4arrow_forward
- What is the expected product for the reaction below?arrow_forwardPropose mechanisms and show the expected products of the following reactions. p@nitrobromobenzene + methylamine (CH3 ¬NH2)arrow_forwardPredict the products of the following reactions. Include stereochemistry when necessary. For reactions with more than one step show the product formed after each step.arrow_forward
