
Concept explainers
(a)
Interpretation:
From
Concept introduction:
By using aufbau principle, electrons starts to occupy from orbitals with lower energy level. Thus, the obtained lower energy configuration is known as ground-state electron configuration.
Aufbau principle:
- Electrons first occupy orbitals with lower energy than higher energy.
- An orbital can be occupied only by two electrons having opposite spins.
- Each electron fills each orbital till it is half filled, when they are degenerate orbital. This is called as Hund’s rule.
- The order of electrons filling in an atom is given as
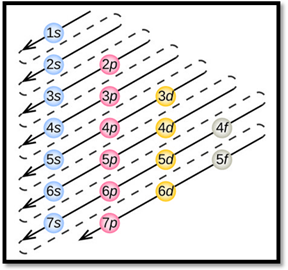
Figure 1
(b)
Interpretation:
From the
Concept introduction:
By using aufbau principle, electrons starts to occupy from orbitals with lower energy level. Thus, the obtained lower energy configuration is known as ground-state electron configuration.
Aufbau principle:
- Electrons first occupy orbitals with lower energy than higher energy.
- An orbital can be occupied only by two electrons having opposite spins.
- Each electron fills each orbital till it is half filled, when they are degenerate orbital. This is called as Hund’s rule.
- The order of electrons filling in an atom is given as
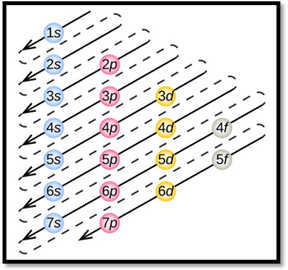
Figure 1
(c)
Interpretation:
From
Concept introduction:
By using aufbau principle, electrons starts to occupy from orbitals with lower energy level. Thus, the obtained lower energy configuration is known as ground-state electron configuration.
Aufbau principle:
- Electrons first occupy orbitals with lower energy than higher energy.
- An orbital can be occupied only by two electrons having opposite spins.
- Each electron fills each orbital till it is half filled, when they are degenerate orbital. This is called as Hund’s rule.
- The order of electrons filling in an atom is given as
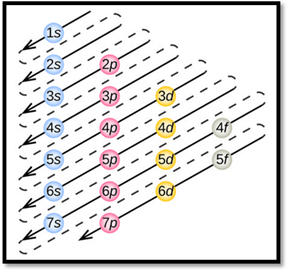
Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
General Chemistry: Atoms First
- Give the ground-state electron configurations of the following elements: (a) P (b) Tc (c) Hoarrow_forwardWhat is the electron configuration of the Ba3+ ion? Suggest a reason why this ion is not normally found in nature.arrow_forwardAn ion with a +4 charge has a ground state electron configuration of [Ar]3d3. What is the ion?arrow_forward
- Give the chemical symbol for the element with the ground‑state electron configuration [Ne]3?23?4[Ne]3s23p4. symbol:arrow_forwardWhat ion with charge +1 has an excited-state electron configuration of [He]2s22p33d1?arrow_forwardExplain, in terms of Coulombic energy and Exchange energy, why the ground-state electron configuration of Cr is [Ar]4s13d5 instead of [Ar]4s23d4.arrow_forward
- Write any two multiplicities of higher energy in the form 2s+1L J, giving a diagrammatic illustration of each.arrow_forwardWhat is the wavelength of the photon absorbed when thehydrogen electron transitions from the n = 1 to the n = 6quantum states?arrow_forwardAtoms of which group in the periodic table have a valence shell electron configuration of ns2?arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

