
Concept explainers
(a)
Interpretation:
The chemical name of
(b)
Interpretation:
The chemical name of
(c)
Interpretation:
The chemical name of
(d)
Interpretation:
The chemical name of
Concept introduction:
Ions contains bunch of atoms having covalent bonds and charged are called polyatomic ions. The name of certain polyatomic ions are given below
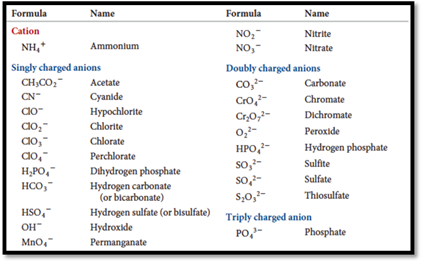
Figure 1
(e)
Interpretation:
The chemical name of
Concept introduction:
Ions contains bunch of atoms having covalent bonds and charged are called polyatomic ions. The name of certain polyatomic ions are given below
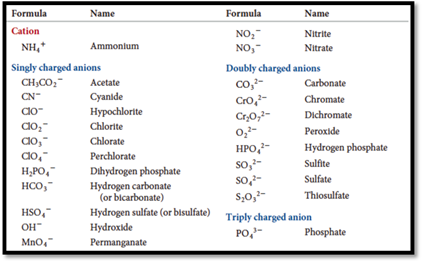
Figure 1
(f)
Interpretation:
The chemical name of
(g)
Interpretation:
The chemical name of
Concept introduction:
Ions contains bunch of atoms having covalent bonds and charged are called polyatomic ions. The name of certain polyatomic ions are given below

Figure 1
(h)
Interpretation:
The chemical name of
Concept introduction:
Ions contains bunch of atoms having covalent bonds and charged are called polyatomic ions. The name of certain polyatomic ions are given below

Figure 1
(i)
Interpretation:
The chemical name of
(j)
Interpretation:
The chemical name of
Concept introduction:
Ions contains bunch of atoms having covalent bonds and charged are called polyatomic ions. The name of certain polyatomic ions are given below

Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
General Chemistry: Atoms First
- I'm having trouble understanding how to put these formulas togetherarrow_forward1.The formula mass of the compound between gallium (III) and oxalate ions in g/mol is: Group of answer choices a, 129.72 b, 495.35 c, 197.82 d, 403.50 2.The formula mass of the compound between barium and phosphate ions in g/mol is: Group of answer choices a, 182.59 b, 601.9 c, 547.77 d, 500arrow_forwardThe mass of a single palladium atom is 1.77×10-22 grams. How many palladium atoms would there be in 91.1 milligrams of palladium?arrow_forward
- 3.1 What is meant by the term molecular mass, and whyis the molecular mass that we calculate generally anaverage molecular mass?arrow_forwardWhich of the following pairs react to form ioniccom-pounds: (a) Cl and Br; (b) Na and Br; (c) P and Se; (d) H and Ba?.arrow_forwardA chemist heats a 6.4 sample of hydrated calcium sulfate, and finds that in the process, 3.4 grams of water vapor is driven off. What is the chemical formula and name of the hydrate?arrow_forward
- Calculate the molecular mass of the following substances: (a) Ibuprofen, C13H18O2 (a drug used as for pain relief)(b) Phenobarbital, C12H12N2O3 (a drug used as a sedative, hypnotic, and antiseizure medication)arrow_forwardDescribe the transformation A to B C to D and D to E Black Balls are carbon, white balls are hydrogen, and green balls are chlorine atoms.arrow_forwardName the following ionic compounds: (a) Li2O, (b) FeCl3,(c) NaClO, (d) CaSO3, (e) Cu1OH22, (f ) Fe1NO322,(g) Ca1CH3COO22, (h) Cr21CO323, (i) K2CrO4, (j) 1NH422SO4.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning




