
General Chemistry: Atoms First
2nd Edition
ISBN: 9780321809261
Author: John E. McMurry, Robert C. Fay
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 3.6, Problem 3.16CP
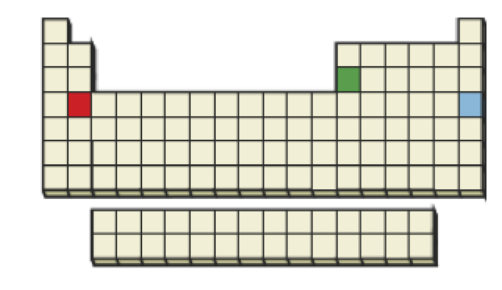
Order the indicated three elements according to the ease with which each is likely to lose its third electron:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
General Chemistry: Atoms First
Ch. 3.1 - Prob. 3.1PCh. 3.1 - Which of the following drawings is most likely to...Ch. 3.2 - Give systematic names for the following compounds:...Ch. 3.2 - Write formulas for the following compounds: (a)...Ch. 3.2 - Prob. 3.5CPCh. 3.2 - Give systematic names for the following compounds:...Ch. 3.2 - Prob. 3.7PCh. 3.2 - Prob. 3.8CPCh. 3.3 - Predict the ground-state electron configuration...Ch. 3.3 - What doubly positive ion has the following...
Ch. 3.4 - Prob. 3.11PCh. 3.4 - which of the following spheres represents a K+...Ch. 3.5 - Using the periodic table as your guide, predict...Ch. 3.6 - (a) Which has the larger third ionization energy,...Ch. 3.6 - Three atoms have the following electron...Ch. 3.6 - Order the indicated three elements according to...Ch. 3.7 - Prob. 3.17PCh. 3.7 - Which of the indicated three elements has the...Ch. 3.8 - What noble-gas configurations are the following...Ch. 3.8 - Prob. 3.20PCh. 3.9 - Calculate the net energy change in kilojoules per...Ch. 3.10 - Which substance in each of the following pairs has...Ch. 3.10 - One of the following pictures represents NaCl and...Ch. 3.11 - Prob. 3.24PCh. 3.11 - Complete the following equations so that the same...Ch. 3.12 - Prob. 3.26PCh. 3.12 - Prob. 3.27PCh. 3.14 - Prob. 3.28PCh. 3 - In the following drawings, red spheres represent...Ch. 3 - Which of the following drawings is more likely to...Ch. 3 - Prob. 3.31CPCh. 3 - Prob. 3.32CPCh. 3 - Prob. 3.33CPCh. 3 - Prob. 3.34CPCh. 3 - Prob. 3.35CPCh. 3 - Prob. 3.36CPCh. 3 - Prob. 3.37CPCh. 3 - Prob. 3.38SPCh. 3 - Prob. 3.39SPCh. 3 - Prob. 3.40SPCh. 3 - Prob. 3.41SPCh. 3 - Prob. 3.42SPCh. 3 - Prob. 3.43SPCh. 3 - Prob. 3.44SPCh. 3 - Prob. 3.45SPCh. 3 - Prob. 3.46SPCh. 3 - Prob. 3.47SPCh. 3 - Prob. 3.48SPCh. 3 - Prob. 3.49SPCh. 3 - Prob. 3.50SPCh. 3 - Prob. 3.51SPCh. 3 - Prob. 3.52SPCh. 3 - What is the identity of the element X in the...Ch. 3 - Prob. 3.54SPCh. 3 - Prob. 3.55SPCh. 3 - Prob. 3.56SPCh. 3 - Prob. 3.57SPCh. 3 - Prob. 3.58SPCh. 3 - Prob. 3.59SPCh. 3 - Prob. 3.60SPCh. 3 - Prob. 3.61SPCh. 3 - Prob. 3.62SPCh. 3 - Prob. 3.63SPCh. 3 - Prob. 3.64SPCh. 3 - Prob. 3.65SPCh. 3 - Prob. 3.66SPCh. 3 - Prob. 3.67SPCh. 3 - Which element in each of the following sets has...Ch. 3 - Prob. 3.69SPCh. 3 - Prob. 3.70SPCh. 3 - Prob. 3.71SPCh. 3 - Prob. 3.72SPCh. 3 - Prob. 3.73SPCh. 3 - Prob. 3.74SPCh. 3 - Prob. 3.75SPCh. 3 - Prob. 3.76SPCh. 3 - Prob. 3.77SPCh. 3 - Prob. 3.78SPCh. 3 - Order the following compounds according to their...Ch. 3 - Calculate the energy change in kilojoules per mole...Ch. 3 - Prob. 3.81SPCh. 3 - Prob. 3.82SPCh. 3 - Prob. 3.83SPCh. 3 - Prob. 3.84SPCh. 3 - Prob. 3.85SPCh. 3 - Calculate the overall energy change in kilojoules...Ch. 3 - The estimated lattice energy for CsF2(s) is +2347...Ch. 3 - Prob. 3.88SPCh. 3 - Prob. 3.89SPCh. 3 - Prob. 3.90SPCh. 3 - Prob. 3.91SPCh. 3 - Prob. 3.92SPCh. 3 - Prob. 3.93SPCh. 3 - Prob. 3.94SPCh. 3 - Prob. 3.95SPCh. 3 - Prob. 3.96SPCh. 3 - Prob. 3.97SPCh. 3 - Prob. 3.98SPCh. 3 - Prob. 3.99SPCh. 3 - Prob. 3.100CHPCh. 3 - Prob. 3.101CHPCh. 3 - Prob. 3.102CHPCh. 3 - Prob. 3.103CHPCh. 3 - Prob. 3.104CHPCh. 3 - Prob. 3.105CHPCh. 3 - Prob. 3.106CHPCh. 3 - Prob. 3.107CHPCh. 3 - Prob. 3.108CHPCh. 3 - Prob. 3.109CHPCh. 3 - Prob. 3.110CHPCh. 3 - Prob. 3.111CHPCh. 3 - Prob. 3.112CHPCh. 3 - Prob. 3.113CHPCh. 3 - Prob. 3.114CHPCh. 3 - Given the following information, construct a...Ch. 3 - Given the following information, construct a...Ch. 3 - Consider the electronic structure of the element...Ch. 3 - Prob. 3.118MPCh. 3 - Prob. 3.119MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 6.84 Which graph correctly depicts the first ionization energy of three elements in groups 14 (dashed line) and 17 (solid line)? Explain the reasoning you used to make your choice.arrow_forwardIn each of the following sets of elements, which element would he expected to have the highest ionization energy? msp;a.Cs,K,Lic.l,Br,Clb.Ba,Sr,Cad.Mg,Si,Sarrow_forwardWithout looking at the figures for the periodic properties, compare the three elements B, AI and C. (a) Place the three elements in order of increasing atomic radius. (b) Rank the elements in order of increasing ionization energy. (c) Which element, B or C, is expected to have the more negative electron attachment enthalpy value?arrow_forward
- Which elements in a given period (horizontal row) of the periodic table lose electrons most easily? Why?arrow_forwardIdentify the atom or ion corresponding to each of the following descriptions: (a) an atom with ground-state electron configuration [Xe]4f145d66s2 (b) an ion with charge 1 and ground-state electron configuration [He]2s22p6 (c) an ion with charge +5 and ground-state electron configuration [Kr]4d6arrow_forwardCompare the elements B, Al, C, Si. (a) Which has the most metallic character? (b) Which has the largest atomic radius? (c) Arrange the three elements B, Al, and C in order of increasing first ionization energy.arrow_forward
- Read the labels of several commercial products and identify monatomic ions of at least four transition elements contained in the products. Write the complete electron configurations of these cations.arrow_forwardive the electron configurations for the following atoms. Do not use the noble gas notation. Write out the complete electron configuration. trong>ElementElectron configuration __________ emsp; __________ emsp; __________ emsp; __________ __________arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
Periodic Properties of Elements | Chemistry | IIT-JEE | NEET | CBSE | Misostudy; Author: Misostudy;https://www.youtube.com/watch?v=L26rRWz4_AI;License: Standard YouTube License, CC-BY
Periodic Trends: Electronegativity, Ionization Energy, Atomic Radius - TUTOR HOTLINE; Author: Melissa Maribel;https://www.youtube.com/watch?v=0h8q1GIQ-H4;License: Standard YouTube License, CC-BY