
Figure 5.32 shows the phase diagram of plagioclase feldspar, which can be considered a mixture of albite
- Suppose you discover a rock in which each plagioclase crystal varies in composition from center to edge, with the centers of the largest crystals composed of 70% anorthite and the outermost parts of all crystal made of essentially pure albite. Explain in some details how this variation might arise. What was the composition of the liquid magma from which the rock formed?
- Suppose you discover another rock body in which the crystals near the top are albite–rich while the crystals near the bottom are anorthite–rich. Explain how this variationmight arise.
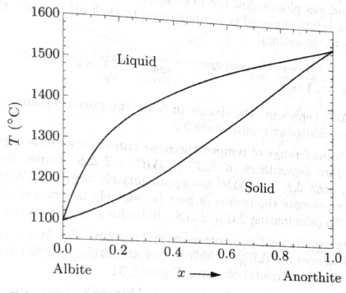
Figure 5.32. The phase diagram of plagioclase feldspar (at atmospheric pressure). From N. L. Bowen, “The Melting Phenomena of the Plagioclase Feldspars,” American Journal of Science 35, 577–599 (1913).
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
An Introduction to Thermal Physics
Additional Science Textbook Solutions
Applied Physics (11th Edition)
Sears And Zemansky's University Physics With Modern Physics
College Physics: A Strategic Approach (4th Edition)
Physics: Principles with Applications
The Cosmic Perspective Fundamentals (2nd Edition)
- Consider the silicon-oxide--silicon structure shown in Fig. 5-41. Both silicon regions are N type with uniform doping of Nd = 1016 cm-3.arrow_forwardIn a gas that is a mixture of water (two H and one O) and carbon dioxide (one C and two O), if the water has average velocity of 327 m/s, what is the average velocity of the carbon dioxide molecule?arrow_forwardAt 100 degree celsius, Copper (Cu) has a lattice length of 3.655 x 10^-10 m. What is its density at the given temperature?arrow_forward
- Use a computer to reproduce the table and graph in Figure 2.4: two Einstein solids, each containing three harmonic oscillators, with a total of six units of energy. Then modify the table and graph to show the case where one Einstein solid contains six harmonic oscillators and the other contains four harmonic oscillators (with the total number of energy units still equal to six). Assuming that all microstates are equally likely, what is the most probable macrostate, and what is its probability? What is the least probable macrostate, and what is its probability?arrow_forwardWould you expect γ to be larger for a gas or a solid? Explain how?arrow_forwardConsider two immiscible liquids such as water and oil. If a spherical oil molecule of radius r is taken out of the oil phase and placed in the water phase, the unfavorable energy of this transfer is proportional to the area of the solute (oil) molecule newly exposed to the solvent (water) multiplied by the interfacial energy, i, of the oil-water interface. The interfacial energy of the bulk cyclohexane-water interface is i = 50 mJ m-2, and the radius of a cyclohexane molecule is 0.28 nm. Using Boltzmann distribution, estimate the solubility of cyclohexane in water at 25 C in units of mol L-1.The concentration of water in water phase is 55.5 mol L-1.arrow_forward
- What properties of a nonideal gas do the van der Waals constants a and b represent?arrow_forwardindicate the following lattice points (211) , (111) and (1 0 1) in an orthorhombic lattice structure with sides a= 5cm,, b =6cm and C =7cmYour answerarrow_forwardDraw the stress-strain diagram of an Al alloy with the yield strength σY=210 MPa, the ultimative tensile strength of UTS=250 MPa and a stress level of σ=160 MPa at fracture (%EL=40%).arrow_forward
- Let's consider the three atoms composing the molecule now have different masses and coordinate, while the axis of rotation is still z axis that is perpendicular to the xy plane. The first atom has a mass of 142.54 kg, with x coordinate at 3 m and y coordinate at 6 m. The second atom has a mass of 82.55 kg, with x coordinate at 1 m and y coordinate at 6 m. The third atom has a mass of 8 kg, with x coordinate at 5 m and y coordinate at 9 m. What is the moment of inertia in unit of kg m2 with respect to the x axis?arrow_forwardWhat evidence can you cite for the microscopic crystal nature of certain solids? For macroscopic crystal nature?arrow_forwardWhy is it valid to represent the structure of a crystalline solid by the structure of its unit cell?arrow_forward
 Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill
Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill
