
Osmotic pressure measurements can be used to determine the molecular weights of large molecules such as protein. For a solution of large molecule to qualify as “dilute,” its molar concentration must be very law and hence the osmotic pressure can be too small to measure accurately. For this reason, the usual procedure is to measure the osmotic pressure at a variety of concentrations, then extrapolate the results to the limit zero concentration. Here are some data,* for the protein hemoglobin dissolved in water at 3°C:
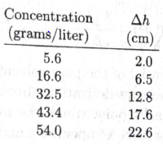
The quantity
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
An Introduction to Thermal Physics
Additional Science Textbook Solutions
Conceptual Physical Science (6th Edition)
College Physics: A Strategic Approach (3rd Edition)
Conceptual Physics (12th Edition)
The Cosmic Perspective Fundamentals (2nd Edition)
College Physics: A Strategic Approach (4th Edition)
College Physics (10th Edition)
- A person is in a closed room (a racquetball court) with v=453 m3 hitting a ball (m 42.0 g) around at random without any pauses. The average kinetic energy of the ball is 2.30 J. (a) What is the average value of vx2 ? Does it matter which direction you take to be x ? (b) Applying the methods of this chapter, find the average pressure on the walls? (c) Aside from the presence of only one "molecule" in this problem, what is the main assumption in Pressure, Temperature, and RMS Speed that does not apply here?arrow_forwardA vertical cylinder of cross-sectional area A is fitted with a tight-fitting, frictionless piston of mass m (Fig. P18.40). The piston is not restricted in its motion in any way and is supported by the gas at pressure P below it. Atmospheric pressure is P0. We wish to find the height h in Figure P18.40. (a) What analysis model is appropriate to describe the piston? (b) Write an appropriate force equation for the piston from this analysis model in terms of P, P0, m, A, and g. (c) Suppose n moles of an ideal gas are in the cylinder at a temperature of T. Substitute for P in your answer to part (b) to find the height h of the piston above the bottom of the cylinder. Figure P18.40arrow_forwardA gas is in a container of volume V0 at pressure P0. It is being pumped out of the container by a piston pump. Each stroke of the piston removes a volume Vs through valve A and then pushes the air out through valve B as shown in Figure P19.74. Derive an expression that relates the pressure Pn of the remaining gas to the number of strokes n that have been applied to the container. FIGURE P19.74arrow_forward
- In the text, it was shown that N/V=2.681025m3 for gas at STP. (a) Show that this quantity is equivalent to N/V=2.681019cm3, as stated. (b) About how many atoms are mere in one m3 (a cubic micrometer) at STP? (c) What does your answer to part (b) imply about the separation of Mama and molecules?arrow_forwardA hand-driven tire pump has a piston with a 3.00 cm diameter and a maximum stroke of 25.0 cm. (a) How much work (in J) do you do in one stroke if the average gauge pressure is 2.40 ✕ 105 N/m2 (about 35 psi)? J (b) What average force (in N) do you exert on the piston, neglecting friction and gravity? Narrow_forwardb. After leaving a vehicle in an indoor car park for a night, you check the tyres’ statistics from the dashboard and gotten the value as follows: Temperature of the air: 24°C Volume of air: 0.0165 m^3 Absolute pressure of the tyre: 3.01 atm Current atmospheric pressure: 1.02 atm To confirm the value, you measure the pressure of the tyre with a gauge pressure and got the value of 1.99 atm. You then drive the car up to Genting Highland where the atmospheric pressure is 0.947 atm due to higher altitude. Once you have reached the destination, the temperature of the air in the tires has now risen to 42.0°C and the volume has risen to 0.0172 m^3. Assume ideal gas in the tyre, calculate the gauge pressure at Genting Highland.arrow_forward
- (A) Prove that the hydrodynamic pressure is equal the kinetic energy per unit volume?arrow_forwardA container with volume 1.63 L is initially evacuated. Then it is filled with 0.226 g of N2. Assume that the pressure of the gas is low enough for the gas to obey the ideal-gas law to a high degree of accuracy.If the root-mean-square speed of the gas molecules is 192 m/s , what is the pressure of the gas? Express the answer in pascals.arrow_forward(a) Calculate the absolute pressure at the bottom of a freshwater lake at a point whose depth is 27.5 m. Assume the density of the water is 1.003X10 kg/m³ and that the air above is at a pressure of 101.3 kPa. (b) What force is exerted by the water on the window of a submarine at this depth if the window is circular and has a diameter of 35 cm?arrow_forward
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
 Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College





