
Concept explainers
Consider the following energy diagram.
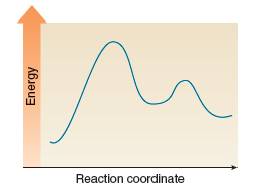
(a) How many steps are involved in this reaction?
(b) Label
(c) Label each transition state.
(d) Which point on the graph corresponds to a reactive intermediate?
(e) Which step is rate-determining?
(f) Is the overall reaction endothermic or exothermic?
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Loose Leaf for Organic Chemistry
Additional Science Textbook Solutions
Introductory Chemistry (6th Edition)
Chemistry: The Central Science (14th Edition)
Chemistry: Structure and Properties
Organic Chemistry
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Principles of Chemistry: A Molecular Approach (3rd Edition)
- Determine rxnH 25 C for the following reaction: NO g O2 g NO2 g This reaction is a major participant in the formation of smog.arrow_forwardxplain why aluminum cans make good storage containers for soft drinks. Styrofoam cups can be used to keep coffee hot and cola cold. How can this be?arrow_forwardConsider the following reaction 3H2(g) + CO2 (g) <=> CH3OH (g) + H2O(g) On the energy diagram, you see two energy curves: with and without catalyst. Chose correct numbers for the following: Energy of the reactant Energy of the products Overall energy of the forward reaction (ΔH) Energy of the transition state without catalyst Activation energy of the forward reaction without catalyst Activation energy of the reverse reaction with catalystarrow_forward
- On an energy diagram of a reaction, the change in enthalpy (ΔH) of a reaction is the difference in energy between __________. the reactants and the products the reactants and the transition state the products and the transition state none of these, it is equal to the activation energyarrow_forwardWhich of the following describes the effect of raising the temperature of an exothermic reaction which has not yet reached equilibrium? a The rate of reaction slows and less product is produced b The rate of reaction slows, but more product is produced c The rate of reaction speeds up, but less product is produced d The rate of reaction speeds up and more product is producedarrow_forwardPlease answer questions 1 to 3 1. What is an exothermic reaction? Draw an energy diagram that illustrates an exothermic reaction. 2. Define catalyst. 3. During an experiment where 50.0 mL of a 1.0 M acid solution was mixed with 50.0 mL of a 1.0 M base solution, the temperature change was measured to be 6.5 ℃. If the density of the resulting mixture is 1.10 g/mL, specific heat of the solution is 4.18 J/g℃, and the Cal constant was 12.0 J/℃, what is the ∆Hrxn in kJ/mol acid?arrow_forward
- 13. Energy is stored in the body in adenosine triphosphate, ATP, which is formed by reaction between adenosine diphosphate, ADP, and dihydrogen phosphate ions. ADP3-(aq) + H2PO42-(aq) -----> ATP4-(aq) + H2O (l) ΔH° = 38 kJ Is the reaction endothermic or exothermic?arrow_forwardIodide, I–, is a catalyst for the decomposition of hydrogen peroxide to give water and oxygen gas: 2 H2O2 (aq) –––> 2 H2O (aq) + O2 (g); ∆Hrxn = –188 kJ/mol Draw a qualitatively accurate reaction diagram (with energy on the y axis, extent of reaction on x axis) to which shows how the path from reactants to products differs between the catalyzed and uncatalyzed versions of the reaction. Label Eain both of the curves. In a few words, define what is meant by “Ea”.arrow_forwardTrue or false? For chemical reactions that occurs in multiple steps, the fastest step in the reaction mechanism requires the most amount of energy to proceed.arrow_forward
- Draw both of these potential energy diagrams labeled with axes, products, reactants,EAarrow_forwardWrite the balanced chemical equation for the conversion of pure magnesium metal to magnesium oxide. Is this reaction reversible or irreversible?arrow_forwardConsider the following energy diagram for the conversion of A → G. a.Which points on the graph correspond to transition states? b. Which points on the graph correspond to reactive intermediates? c.How many steps are present in the reaction mechanism? d. Label each step of the mechanism as endothermic or exothermic. e.Label the overall reaction as endothermic or exothermic.arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,


