
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 7, Problem 16CTQ
Interpretation Introduction
Interpretation: In the given structures, each black ball needs to be labeled as up or down.
Concept Introduction: Chair conformation is the most stable conformation of cyclohexane. It is represented as follows:

Here, the substituted groups in the chair conformation are represented as follows:
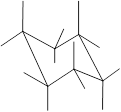
The groups showing in straight upward and downward directions are axial and the groups bend slightly right or left are equatorial.
The axial and equatorial groups are represented in the chair conformation as A and E:
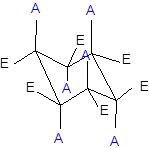
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Circle and name three functional groups within the molecule and place an * against any chiral carbons
Please help with row 2/question 2 ; the instructions for the specific column in row 2 are above row 1:
Follow the instructions in each column. Hint for the last column: draw it with a wedge and again with adash – which gives the correct configuration?
hey could someone help me label all of the chiral carbons as R or S? Thanks!
Chapter 7 Solutions
Organic Chemistry: A Guided Inquiry
Ch. 7 - Prob. 1CTQCh. 7 - Prob. 2CTQCh. 7 - Prob. 3CTQCh. 7 - Draw wedge and dash skeletal representations of...Ch. 7 - Label each ring in Figure 7.2 cis or trans.Ch. 7 - Prob. 6CTQCh. 7 - Prob. 7CTQCh. 7 - Prob. 8CTQCh. 7 - a model of cyclohexane in a chair conformation,...Ch. 7 - Prob. 10CTQ
Ch. 7 - Prob. 11CTQCh. 7 - Fill in the blanks: cis-1,3-Dimethylcyclohexane...Ch. 7 - Prob. 13CTQCh. 7 - Prob. 14CTQCh. 7 - Prob. 15CTQCh. 7 - Prob. 16CTQCh. 7 - Prob. 17CTQCh. 7 - Prob. 18CTQCh. 7 - Draw chair representations of...Ch. 7 - Which stereoisomer in the previous question is...Ch. 7 - Prob. 21CTQCh. 7 - Prob. 1ECh. 7 - Label each of the following as cis, trans or...Ch. 7 - Which pair has more in common with one another?Ch. 7 - Prob. 4ECh. 7 - Prob. 5ECh. 7 - Prob. 6ECh. 7 - Fill in the table by drawing a representation of a...Ch. 7 - Prob. 9ECh. 7 - True or False: If you perform a chair flip on...Ch. 7 - Prob. 11ECh. 7 - Prob. 12ECh. 7 - Prob. 13ECh. 7 - Prob. 14ECh. 7 - Prob. 15ECh. 7 - Draw trans-1-tert-butyl-3-methylcyclohexane in its...Ch. 7 - Build a model of methylcyclohexane, and use the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- I know the answer is correct but I can’t figure out if C is tertiary ? It can’t be secondary because it has more than two carbons connected to the +Carrow_forwardShown below is Streptomycin, and Neomycin B. Circle and label as many functional groups in these molecules as you can. a. Label each chiral carbon in Streptomycin. How many total stereoisomers exist for Streptomycin? b. Label each chiral carbon in Neomycin B. How many total stereoisomers exist for Neomycin B?arrow_forwardUse Model 1 to propose names for three-, four-, five-, and six-carbon branches that follow the same pattern as “methyl” and “ethyl” for one- and two-carbon branches, respectively. (Note: The names of seven-, eight-, etc. carbon branches follow the same pattern, but branches of such length are rare since they are usually the parent chain.)arrow_forward
- (E) Write a correct name below each of the unbranched alkanes in CTQ 1.arrow_forwardcircle and name each fundamental group present in the following moleculearrow_forwardConstruct a model in which a tetrahedral carbon atom has four different colored model atoms attached to it- red, green, orange and white representing 4 different atoms attached to the central atom. a) Does the atom have a plane of symmetry? why or why not? b) Now replace the green atom in your model with a second orange atom. Now two of the groups attached to the carbon atom are identical. Does the model now have a plane of symmetry? Describe it. c)A carbon atom has four different groups attached to the stereogenic center. Draw structural formulas for the following compound and mark stereogenic centers with as asterisk: 1-bromobutane, 2-bromobutane, 1,2-dibromobutane, 1,4-dibromobutane, 2,3-dibromobutane.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY