
Concept explainers
(a)
Interpretation:
Molecular shape and bond angle of A-B needs to be explained.
Concept introduction:
A molecule’s physical and chemical properties are determined through its shape. The shapes of the reactant molecules play an important role in determining whether they come close to react or not. The densities of electrons generated by the overlap of the mutual electrons' orbitals define the molecular structure.
(a)
Answer to Problem 108A
The shape of the molecule A-B is linear and its bond angle is
Explanation of Solution
The A-B molecule contains one bonding pair. Hence, its molecular shape is linear and its bond angle is
(b)
Interpretation:
Molecular shape and bond angle of A-B-A needs to be explained.
Concept introduction:
A molecules physical and chemical properties are determines through its shape. The shapes of the reactant molecules play an important role in determining whether they come close to react or not. The densities of electrons generated by the overlap of the mutual electrons' orbitals define the molecular structure.
(b)
Answer to Problem 108A
The shape of the molecule A-B-B is linear and its bond angle is
Explanation of Solution
The A-B-A molecule contains two bonding pair. Hence, its molecular shape is linear and its bond angle is
(c)
Interpretation:
Molecular shape and bond angle of following molecules needs to be explained.
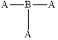
Concept introduction:
A molecules physical and chemical properties are determines through its shape. The shapes of the reactant molecules play an important role in determining whether they come close to react or not. The densities of electrons generated by the overlap of the mutual electrons' orbitals define the molecular structure.
(c)
Answer to Problem 108A
The shape of the molecule is trigonal planar and its bond angle is
Explanation of Solution
The molecule contains three pair of bonding electrons. Hence, its molecular shape is trigonal planar and its bond angle is
(d)
Interpretation:
Molecular shape and bond angle of following molecule needs to be explained.
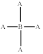
Concept introduction:
A molecules physical and chemical properties are determines through its shape. The shapes of the reactant molecules play an important role in determining whether they come close to react or not. The densities of electrons generated by the overlap of the mutual electrons' orbitals define the molecular structure.
(d)
Answer to Problem 108A
The shape of the molecule is tetrahedral and its bond angle is
Explanation of Solution
The molecule contains four bonding pairs. Hence, its molecular shape is tetrahedral and its bond angle is
Chapter 8 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
Chemistry: Structure and Properties
Chemistry: The Central Science (14th Edition)
CHEMISTRY-TEXT
Inorganic Chemistry
Organic Chemistry (8th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





