
Concept explainers
Interpretation:
The Lewis structure of ethylene glycol is to be drawn. The sigma and pi bonds in ethylene glycol are to be identified.
Concept introduction:
The strategy for drawing Lewis structure is mention below.
- Calculate the number of valence electrons present in the molecule.
- Calculate the electron pairs by diving number of valence electrons by 2.
- Determine the bond pairs.
- Determine the lone pairs.
- Check whether the central atom satisfies octet rule.
Answer to Problem 141A
The Lewis structure of ethylene glycol is drawn below.
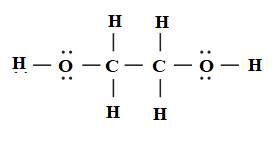
All the bonds in ethylene glycol are sigma bond. Due to the miscibility of ethylene glycol in water, the freezing point of water decreases. Therefore, ethylene glycol acts as a antifreeze-coolant.
Explanation of Solution
The chemical formula of ethylene glycol is
The number of electron pair is calculated as shown below.
Among thirteen electron pairs, nine electron pairs are involved in bonding and four electron pairs remain as lone pairs.
Therefore, the Lewis structure of ethylene glycol is drawn below.
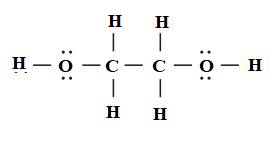
It is known that in single bond only sigma bond is present. In double bond, one bond is sigma and one is pi bond. In triple bond, one bond is sigma and the remaining two bonds are pi bond. The lone pairs are non bonding electrons represented by two dots.
All the bonds present in ethylene glycol are single bond. Therefore, all the bonds in ethylene glycol are sigma bond.
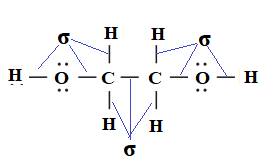
Due to presence of hydroxyl group
Chapter 8 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
Introductory Chemistry (5th Edition) (Standalone Book)
Essential Organic Chemistry (3rd Edition)
Organic Chemistry
Inorganic Chemistry
General Chemistry: Principles and Modern Applications (11th Edition)
Chemistry: The Central Science (14th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





