
EBK LIFE: THE SCIENCE OF BIOLOGY
11th Edition
ISBN: 8220103935432
Author: Sadava
Publisher: MAC HIGHER
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 9.3, Problem 4R
Summary Introduction
To review:
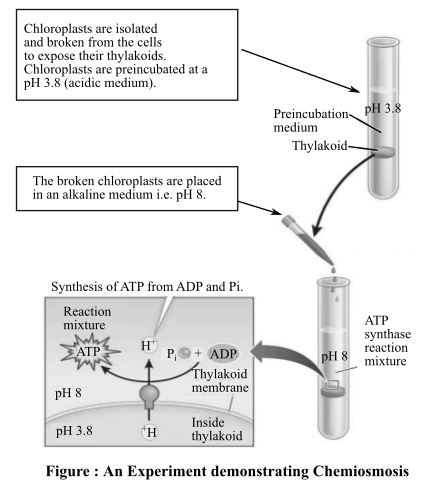
The consequences of experiment in figure 9.9 in the presence of antimycin A drug.
Introduction:
Figure shows an experiment, which was performed to prove that a proton concentration gradient drove formation of adenosine triphosphate (ATP) in the chloroplasts. Chemiosmosis occurs after electron transport chain (ETC) occurs inside mitochondria, in which production of ATP takes place by influx of protons back into the matrix of mitochondria.
The antimycin A drug is known to block electron transport in the chloroplasts and mitochondria.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Compare the abundance of cristae in the mitochondria in Fig. 18.11b (677) with those in Fig. 20.21 (760). Is there a difference? What might this reflect in functional terms?
waste product of anaerobic respiration by animal cells
seous product of the Krebs cycle
udent set up the apparatus shown below, one with germinating seeds and one with dry seeds. The
ssium hydroxide (KOH) in each tube will absorb any carbon dioxide in the test tube so that carbo
ide released will not affect the volume of gas in the test tube. This ensures that the volume of
gen gas in the test tube is the variable being measured.
student left the apparatus for 30 minutes in a warm, dark room and then measured the location c
droplet of dye. What are the expected results?
t tube A
dry seeds
-KOH
d
Test tube B
Time left 1:01
germinating seeds
-KOH
lect one:
a. In test tube B, the droplet moved to the left, indicating that oxygen was used by the germinatim
seeds as they were growing.
b. In test tube the droplet moved to the left, indicating that oxygen was released by the dry
seeds.
40
Oc. In test tube B, the droplet moved to the left, indicating that oxygen was released by the
germinating…
If you examine OX/PHOS in the organism that has the c16 ATP Synthase complex from Q3, how many ATPs will be generated from one Succinate?# of ATPs:
Chapter 9 Solutions
EBK LIFE: THE SCIENCE OF BIOLOGY
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- CYP2E1 has a much higher Km for ethanol than ADH (11 mM versus 0.05 mM,respectively). When greater portions of ethanol are ingested (0.08% or 17 mM), which part of the statement would be true?arrow_forwardAbout the process of industrial production of ethanol by the yeast Saccharomyces cerevisiae, mark the correct alternatives: (a) the cells must be cultured in anaerobic conditions to activate the metabolic pathway of ethanol production (b). the presence of oxygen is required to allow regeneration of the NAD+ cofactor (c). good oxygenation of the medium is important to favor the formation of greater amounts of ATP (d).the production of ethanol is always accompanied by the formation of glycerol (e). the sugar present in the culture medium is completely oxidized to CO2 and waterarrow_forwardPlace numbers beside the statements below to indicate the order of events occurring in the light reactions. I know for sure that 1 and 8 are correct, but the rest I’m not too confident about. I attached both my work and a blank copy.arrow_forward
- Write the steps for Krebs Cycle Pathwayarrow_forwardWhen pure reduced cytochrome c is added to carefully prepared mito- chondria along with ADP, P, antimycin A, and oxygen, the cytochrome c becomes oxidized, and ATP is formed, with a P/O ratio approaching 1. Why was antimycin A added, and what does this experiment tell you about the location of coupling sites?arrow_forwardShown in the figure, is a portion of the electron transport chain pathway, in which electrons are transferred from NADH to O2, the energy of this reaction is coupled to the pumping of H+. Drug X can pick up electrons from specific intermediates as shown in the figure. You treat cells carrying out respiration with a saturating dose of drug X, so that all the electrons which would normally continue in the pathway are captured by drug X. What would you expect in these cells? Select all that apply. 4 Drug X e- A NADH + H+ Electron transport H₂O e- Proton pumping 0₂ NAD+ H* (space A) H* (space B) The cells continue to produce carbon dioxide The cells do not produce carbon dioxide Rate of ATP synthesis decreases compared to normal Rate of ATP synthesis increases compared to normalarrow_forward
- The reduction potentials of iron in each of the cytochromes in the electron transport complexesvary from 0.The reduction potentials of iron in each of the cytochromes in the electron transport complexes vary from 0.1 V to 0.39 V. Explain why these different values are necessary for the operation of this processarrow_forwardIn an experiment, mitochondria were suspended in a buffered medium. The O2 electrode was used to monitor 0, consumption and assayed for the presence of ATP. Figure 3 shows the respiratory control by following the oxygen utilization (solid line) and ATP synthesis (discontinuous line) in the isolated mitochondria with additions of succinate, ADP, orthophosphate, oligomycin and dinitrophenol (DNP). Comment on the observations and relate the effects of the compounds added to electron transport and oxidative phosphorylation. Add DNP Add oligomycin Add ADP + P Add succinate Time Figure 3 Og consumed ATP synthesizedarrow_forward13arrow_forward
- 12. Use Figure 4 for questions a-c below. 114 ATP synthase matrix intermembrane space Figure 4: Sketch of a mitochondrion https://upload.wikimedia.org/wikipedia/commons/1/1a/Schema mitochondrion basic.svg Bionet, CC BY-SA 4.0 , via Wikimedia Commons a. Add to the sketch to show a H* concentration gradient in an active mitochondrion. (Sketch more H* in the appropriate space. Sketch less H* in the appropriate space.) 4+ b. Sketch an arrow to represent the direction that protons flow (via diffusion) through ATP synthase. c. If the concentration of protons was equal on each side of the membrane, how would that affect the flow of protons through ATP synthase? How would that affect ATP production from glucose?arrow_forwardA newly identified bacterium is unable to synthesize ubiquinone. A mobile electron carrier called CXC3 is used as a substitute. From the information provided in the table, calculate the delta G knot prime and the Keq value at 298 K for the redox reaction that occurs in this bacterium’s electron transport chain. Explain the impact that using CXC 3 instead of ubiquinol will have on ATP production in the cell. How might this cell adapt to this situation?arrow_forwardWhich of the following statements concerning the complete oxidation of FADH2 in the electron transport chain is NOT true? a. In the final step, electrons from cytochrome c to O2 reducing it to H2O in complex IV, and four protons are transported from the intermembrane space to the matrix. b. In the first step, electrons from FADH2 are transferred in complex II to ubiquinone, which does not transport any proton across the inner mitochondrion membrane. c. In the second step, complex III transfers the electrons from ubiquinone to cytochrome c, and four protons are transported from the matrix to the intermembrane space. d. The complete oxidation of FADH2 causes transfer of 6 protons and yields two ATP.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning

Biology Today and Tomorrow without Physiology (Mi...
Biology
ISBN:9781305117396
Author:Cecie Starr, Christine Evers, Lisa Starr
Publisher:Cengage Learning