
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 12.17SP
Predict the masses and the structures of the most abundant fragments observed in the mass spectra of the following compounds.
- a. 2-methylpentane
- b. 3-methylhex-2-ene
- c. 4-methylpentan-2-ol
- d. 2-methyl-1-phenylpropane
- e. cyclohexyl isopropyl ether [cyclohexyl—O—CH(CH3)2]
- f. CH3CH2CH2NHC(CH3)3 tert-butylpropylamine
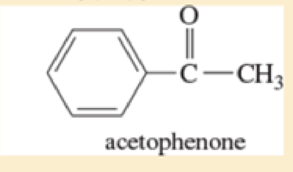
- g. *3-bromo-2-methylpentane
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Predict the masses and the structures of the most abundant fragments observed in the mass spectra of the following compounds.
(a) 4-methylpentan-2-ol (b) 2-methyl-1-phenylpropane
Predict the masses and the structures of the most abundant fragments observed in the mass spectra of the followingcompounds.(a) 2-methylpentane (b) 3-methylhex-2-ene (c) 4-methylpentan-2-ol(d) 2-methyl-1-phenylpropane (e) cyclohexyl isopropyl ether [cyclohexyl¬O¬CH(CH3)2](f) CH3CH2CH2NHC(CH3)3tert@butyl propyl amine(g) C CH3Oacetophenone(g) *(h) 3-bromo-2-methylpentane
Predict the masses and the structures of the most abundant fragments observed in the mass spectra of the followingcompounds.(a) 2-methylpentane (b) 3-methylhex-2-ene (c) 4-methylpentan-2-ol(d) 2-methyl-1-phenylpropane
Chapter 12 Solutions
Organic Chemistry (9th Edition)
Ch. 12.3 - Complete the following conversion table. (cm1)...Ch. 12.5 - Which of the bonds shown in red are expected to...Ch. 12.7C - For each hydrocarbon spectrum, determine whether...Ch. 12.9A - Spectra are given for three compounds. Each...Ch. 12.10 - The infrared spectra for three compounds are...Ch. 12.12 - Prob. 12.6PCh. 12.14B - Identify which of these four mass spectra indicate...Ch. 12.15A - Show the fragmentation that accounts for the...Ch. 12.15A - Show the fragmentations that give rise to the...Ch. 12.15B - Ethers are not easily differentiated by their...
Ch. 12.15C - Prob. 12.11PCh. 12 - Prob. 12.12SPCh. 12 - Prob. 12.13SPCh. 12 - All of the following compounds absorb infrared...Ch. 12 - Prob. 12.15SPCh. 12 - Four infrared spectra are shown, corresponding to...Ch. 12 - Predict the masses and the structures of the most...Ch. 12 - Prob. 12.18SPCh. 12 - Prob. 12.19SPCh. 12 - (A true story) While organizing the undergraduate...Ch. 12 - Prob. 12.21SPCh. 12 - Prob. 12.22SPCh. 12 - An unknown, foul-smelling hydrocarbon gives the...Ch. 12 - covered a synthesis of alkynes by a double...Ch. 12 - Three IR spectra are shown, corresponding to three...Ch. 12 - Prob. 12.26SPCh. 12 - Prob. 12.27SPCh. 12 - Prob. 12.28SPCh. 12 - The ultimate test of fluency in MS and IR is...Ch. 12 - Prob. 12.30SPCh. 12 - Consider the following four structures, followed...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Determine the de Brogue wavelength of a. an electron moving at 1/10 the speed of light. b. a 400 g Frisbee movi...
Inorganic Chemistry
Problem 11.1 Neopheliosyne B is a novel acetylenic fatty acid isolated from a New Caledonian marine sponge. (a)...
Organic Chemistry
Characterize each of the following structures as aromatic, nonaromatic, or antiaromatic:
Answer: _____
Organic Chemistry As a Second Language: Second Semester Topics
Fully developed conditions are known to exist for water flowing through a 25-nim-diameer tube at 0.01 kg/s and ...
Fundamentals of Heat and Mass Transfer
Determine the number of protons, neutrons, and electrons in the following atoms: a. a hydrogen atom that has a ...
General, Organic, and Biological Chemistry (3rd Edition)
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living By Chemistry: First Edition Textbook
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Predict the masses and the structures of the most abundant fragments observed in the mass spectra of the following compounds. (a) acetophenone (b) 3-bromo-2-methylpentanearrow_forwardExplain per step. The mass spectra of 1-methoxybutane, 2-methoxybutane, and 2-methoxy-2-methylpropaneare shown. Match the compounds with the spectra.arrow_forwardPredict the masses and the structures of the most abundant fragments observed in the mass spectra of the followingcompounds. cyclohexyl isopropyl ether [cyclohexyl¬O¬CH(CH3)2]arrow_forward
- Can someone go step by step on how to predict the masses and structure of the most abundant fragments of the 3-methylhex-2-ene mass spectra?arrow_forwardPredict the masses and the structures of the most abundant fragments observed in the mass spectra of the following compounds. (a) cyclohexyl isopropyl ether (b) tert-butyl propyl aminearrow_forwardAccount for the formation of the base peak in these mass spectra. ( Q.) Isobutylamine, m/z 30arrow_forward
- 1. Suggest a way of distinguishing between pentan-2-one and pentan-3-one (CH3COCH2CH2CH3 and CH3CH2COCH2CH3) using their mass spectra with proper figures (Spectra) and clear fragmented ions.arrow_forwardWhich of the following molecules matches the given data? (Note: All the molecules have a M+ = m/z 102) C5H10O2 base peak = m/z 43arrow_forwardDraw the structures of the fragments that give rise to the peaks at m/z 43, 57 and 85 in the mass spectrum of 2,4 - dimethylpentane. Thank you!arrow_forward
- Why does the mass spectrum of 2-Methyl-2-hexanol is different from the M+1 peak from the mass spectrum?arrow_forwardEI-MS of ortho nitrotoluene shows a large fragment ion at m/z 120 .The EI-MS of Tri deuteron ortho nitrotoluene does not show any peak at m/z 120 but does have a peak at m/z 122 .Show the fragmentation pattern that accounts for the above observation.arrow_forwardAccount for the formation of the base peaks in these mass spectra. Q.) Isobutylmethylamine, m/z 44arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY