
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12, Problem 12.20SP
(A true story) While organizing the undergraduate stockroom a new chemistry professor found a half-gallon jug containing a cloudy liquid (bp 100-105 °C), marked only “STUDENT PREP.” She ran a quick mass spectrum, which is printed below. As soon as she saw the spectrum (without even checking the actual mass numbers), she said, “I know what it is.”
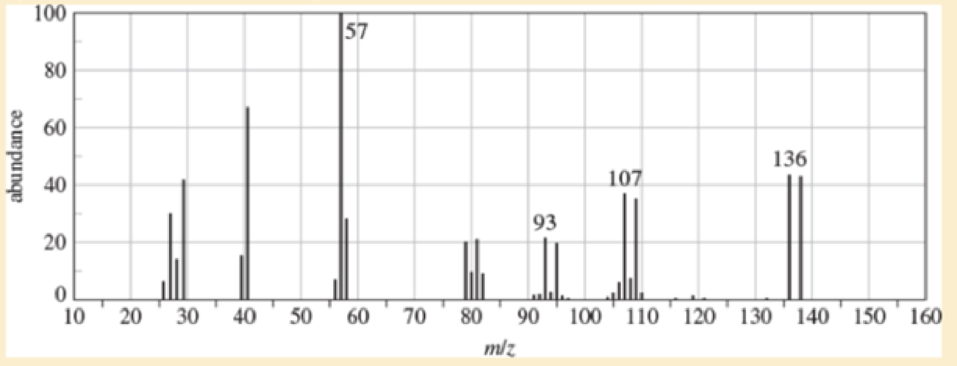
- a. What compound is the “student prep”? Any uncertainty in the structure?
- b. Suggest structures for the fragments at 136, 107, and 93. Why is the base peak (at m/z 57) so strong?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Mass Spec:How could you distinguish the mass spectrum of 2,2-dimethylpropane from those of pentane and 2-methylbutane in a mass spectrometer?
Using curved arrows, show the principal fragments you would expect to see in the mass spectrum of each of the following compounds:
What is the molecular formula of the attached image?
Chapter 12 Solutions
Organic Chemistry (9th Edition)
Ch. 12.3 - Complete the following conversion table. (cm1)...Ch. 12.5 - Which of the bonds shown in red are expected to...Ch. 12.7C - For each hydrocarbon spectrum, determine whether...Ch. 12.9A - Spectra are given for three compounds. Each...Ch. 12.10 - The infrared spectra for three compounds are...Ch. 12.12 - Prob. 12.6PCh. 12.14B - Identify which of these four mass spectra indicate...Ch. 12.15A - Show the fragmentation that accounts for the...Ch. 12.15A - Show the fragmentations that give rise to the...Ch. 12.15B - Ethers are not easily differentiated by their...
Ch. 12.15C - Prob. 12.11PCh. 12 - Prob. 12.12SPCh. 12 - Prob. 12.13SPCh. 12 - All of the following compounds absorb infrared...Ch. 12 - Prob. 12.15SPCh. 12 - Four infrared spectra are shown, corresponding to...Ch. 12 - Predict the masses and the structures of the most...Ch. 12 - Prob. 12.18SPCh. 12 - Prob. 12.19SPCh. 12 - (A true story) While organizing the undergraduate...Ch. 12 - Prob. 12.21SPCh. 12 - Prob. 12.22SPCh. 12 - An unknown, foul-smelling hydrocarbon gives the...Ch. 12 - covered a synthesis of alkynes by a double...Ch. 12 - Three IR spectra are shown, corresponding to three...Ch. 12 - Prob. 12.26SPCh. 12 - Prob. 12.27SPCh. 12 - Prob. 12.28SPCh. 12 - The ultimate test of fluency in MS and IR is...Ch. 12 - Prob. 12.30SPCh. 12 - Consider the following four structures, followed...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (14th Edition)
Practice Problem ATTEMPT
Write the rate expressions for each of the following reactions:
(a)
(b)
(c)
Chemistry
Give the IUPAC name for each compound.
Organic Chemistry
Real walls are never totally adiabatic. Use your experience to order the following walls in increasing order wi...
Thermodynamics, Statistical Thermodynamics, & Kinetics
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which one is the incorrect statement regarding mass spectrum? Question 25 options: In case of alcohols, M-18 peak is produced due to the loss of neutral H2O molecule (Dehydration) The relative heights of (M)+• peak and (M+1)+• peak is used to calculate the number of carbon atoms The structure likely contains a bromine atom if the (M+2)+• peak and (M)+• are almost similar in heights The molecular ion peak (M)+• always represent the base peak The structure likely contains a chlorine atom if the (M+2)+• peak is approximately one-third as tall as (M)+•arrow_forwardA) Label the mass spectrum of 1-butanol,3-methyl (Be sure to write the chemical formula for each major peak) (major peaks include 88, 70, 55, 42, and 43 m/z)arrow_forward. A compound made of C, H, and Cl shows two peaks on a mass spectrum, one at 52 u and the other at 50 u. What is a reasonable molecular formula for this compound? Assume that there are only 1H, 12C, 35Cl and37Cl in the compoundarrow_forward
- Chemistry Name the compound using mass spec, NMR, and IR.arrow_forwardQ2. Write appropriate answer. ( Analysis of an unknown compounds shows its molecular weight 155. Elemental analysis shows that it contains C (46.45%), hydrogen H (5.85%), N (27.08%) and O (20.62%). Using the rule of thirteen, calculate the molecular formula and index of hydrogen deficiency for the compound. 2arrow_forwardWhich one is the incorrect statement regarding mass spectrum? options: The structure likely contains a chlorine atom if the (M+2)+• peak is approximately one-third as tall as (M)+• The relative heights of (M)+• peak and (M+1)+• peak indicate the number of carbon atoms A signal at M-15 indicates the loss of a methyl group and a signal at M-29 indicates the loss of an ethyl group The structure likely contains a bromine atom if the (M+2)+• peak and (M)+• are almost similar in heights In case of alcohols, a signal at m/z = 18 is produced due to the loss of neutral H2O molecule (Dehydration)arrow_forward
- Which of the following compounds gives this mass spectrum and explain why it is so?arrow_forwardHow would you use mass spectrometry to distinguish between chloroform (CHCl3) and deutorated chloroform (CDCl3)?arrow_forwardWhich of the following compounds is most likely to correspond to the mass spectrum below? Provide explanations for your choice.arrow_forward
- For which compound containing a heteroatom (an atom other than carbon or hydrogen) does the molecular ion have an even-numbered mass? For which does it have an odd-numbered mass? Q.)A thiol with the molecular formula CnH2n+1SHarrow_forwardPhencyclidine is c17h25n a same suspected of being the illicit drug was formed to be 84.71 c 10.42h and 5.61 n do these data acceptably match the theoretical data for phencyclidinearrow_forwardHi, I need help balancing these equations. CCl + K2CrO4 ==> Cl2CO + CrO2Cl2 + KCl & K2CrO4 + C ==> K2CO3 + K2O + CO + Cr2O3 Thank you!!!arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY