
Concept explainers
Write a balanced equation for the combustion of each sycloalkane.
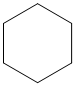
(a)
Interpretation:
The balance equation for the combustion of following cycloalkane should be written.
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
Saturated hydrocarbon is known as alkane having general molecular formula
The compounds in which series of atoms are connected to form a ring is known as cyclic compound whereas the compounds which are open chain compounds and their atoms doesn't form a ring is known as acyclic compounds.
The chemical reaction which involves the reaction of alkane and oxygen, results in the formation of carbon dioxide gas and water vapor is known as combustion reaction. The combustion reaction is an exothermic reaction.
The general reaction of combustion of alkane is:
A reaction is said to be balanced, if the number of atoms of each element on the product side and one the reactant side of a chemical reaction are equal.
Answer to Problem 66P
Explanation of Solution
Cyclic alkane is defined as an alkane in which series of atoms are combined to form a ring.
The general molecular formula of cyclic alkane is
The given cycloalkane is:
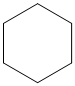
The chemical formula of the cycloalkane is
The combustion reaction of
Reactant side Product side
Number of C atoms = 6 Number of C atoms = 1
Number of H atoms = 12 Number of H atoms = 2
Number of O atoms = 2 Number of O atoms = 3
Thus, the above reaction is not balanced. To balance the reaction, multiply 9 with oxygen gas, 6 with carbon dioxide gas and 6 with water vapor.
Therefore, the balanced combustion reaction of
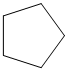
(b)
Interpretation:
The balance equation for the combustion of following cycloalkane should be written.
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
Saturated hydrocarbon is known as alkane having general molecular formula
The compounds in which series of atoms are connected to form a ring is known as cyclic compound whereas the compounds which are open chain compounds and their atoms doesn't form a ring is known as acyclic compounds.
The chemical reaction which involves the reaction of alkane and oxygen, results in the formation of carbon dioxide gas and water vapor is known as combustion reaction. The combustion reaction is an exothermic reaction.
The general reaction of combustion of alkane is:
A reaction is said to be balanced, if the number of atoms of each element on the product side and one the reactant side of a chemical reaction are equal.
Answer to Problem 66P
Explanation of Solution
Cyclic alkane is defined as an alkane in which series of atoms are combined to form a ring.
The general molecular formula of cyclic alkane is
The given cycloalkane is:
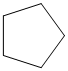
The chemical formula of the cycloalkane is
The combustion reaction of
Reactant side Product side
Number of C atoms = 5 Number of C atoms = 1
Number of H atoms = 10 Number of H atoms = 2
Number of O atoms = 2 Number of O atoms = 3
Thus, the above reaction is not balanced. To balance the reaction, multiply
Therefore, the balanced combustion reaction of
Want to see more full solutions like this?
Chapter 12 Solutions
General, Organic, and Biological Chemistry - 4th edition
- How does the structure of a cycloalkane differ from that of a straight-chain or branched-chain alkane?arrow_forwardWhat is the difference in bonding and in general molecular formula between an alkene and a cycloalkane with the same number of carbon atoms?arrow_forwardWrite molecular formulas for each bicycloalkane, given its number of carbon atoms. Q.) Hydrindane (9 carbons)arrow_forward
- Write molecular formulas for each bicycloalkane, given its number of carbon atoms. Q.) Decalin (10 carbons)arrow_forwardWhat is the name of the cycloalkane containing five carbon atoms? A) cyclohexane B) cyclobutane C) cyclopentane D) cyclic pentane E) cyclic hexanearrow_forwardWrite balanced equations for the complete combustion of propane and methylcyclopentane.arrow_forward
- Classify/ Name the cycloalkanes below:arrow_forwardWrite a complete balanced chemical equation using structural formulas to show how bromocyclohexane can be made from a cycloalkane. Include all products made. Name all reactants and the type of reaction that occurs.arrow_forwardDefine Cyclohexane ?arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning



