
(a)
Interpretation:
The IUPAC name of the following alkane should be determined.
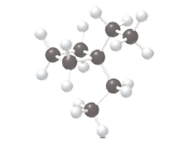
Concept Introduction:
In order to give an IUPAC name to an alkane following rules are followed:
- The longest carbon chain that is parent chain is determined and suffix −ane for alkane is used in the name.
- For cyclic alkanes, the prefix cyclo is used in the name to indicate that the alkane is cyclic.
- The numbering of the parent chain is done in such a way that the substituents get the lowest numbers.
- Name should be written in alphabetical order.
- Hyphen is used to connect the number to the name.
For number of carbons atoms in alkane chain, the prefix is given as:
Carbon-1 meth
Carbon-2 eth
Carbon-3 prop
Carbon-4 but
Carbon-5 pent
Carbon-6 hex
Carbon-7 hept
Carbon-8 oct
Carbon-9 non
Carbon-10 dec
(b)
Interpretation:
One constitutional isomer of the following alkane should be drawn.
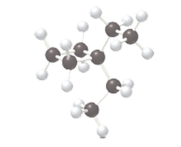
Concept Introduction:
Constitutional isomers can be defined as the pair of molecules with the same molecular formula, but different structural formula. This phenomenon is known as isomerism.
(c)
Interpretation:
The solubility of given molecule in water should be explained.
Concept Introduction:
The solubility of a molecule depends on the polarity of the solvent and solution that means like dissolve like. In other words, the polar molecule is soluble in polar solvent only and in non-polar solvents, the soluble molecule will be non-polar in nature.
(d)
Interpretation:
The solubility of given molecule in organic solvent should be explained.
Concept Introduction:
The solubility of a molecule depends on the polarity of the solvent and solution that means like dissolve like. In other words, the polar molecule is soluble in polar solvent only and in non-polar solvents, the soluble molecule will be non-polar in nature.
(e)
Interpretation:
The balanced equation for the complete combustion of the given compound should be determined.
Concept Introduction:
Chemical equation is written in such a way that the symbolic representation of reaction represents the reaction taking place in the system. The reactants are written on the left-hand side and the products are written on the right-hand side of the equation and are separated by an arrow, two or more reactants and products are separated by "+".
The reactions for those the number of atoms of each element in the reactant and in the product, side are equal, such reactions are said to be a balanced chemical equation.
(f)
Interpretation:
The skeletal structure of the given compound should be determined.
Concept Introduction:
The formula of a compound that shows the bonds between the atoms and other atoms omitting carbon and hydrogen atoms is said to be skeletal structure of that compound. In these structures, the bond between any atom(s) and hydrogen is also omitted.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
General, Organic, and Biological Chemistry - 4th edition
- How To Name a Cycloalkane Using the IUPAC System ?arrow_forwardExplain why diethyl ether's boiling point is lower than the boiling point of butan-1-ol. Support your answer using the concept of intermolecular forces of attraction.arrow_forwardwhat is the full iupac name include isomerismarrow_forward
- why boiling point of alcohols is more then the corresponding aliphatic hydrocarbons?arrow_forwardWhen the molecular formulas for cyclic and noncyclic alkanes with the same number of carbon atoms are compared, it is always found that the cycloalkane has a) two more hydrogen atoms. b) the same number of hydrogen atoms. c) two less hydrogen atoms. d) four less hydrogen atoms.arrow_forwardAnswer the following question about the alkane drawn below. CH3 CH2CH3 | | CH3CH2CH2 ―C― C― CH2CH3 | | CH3 H Write a balanced equation for complete combustion (do not include states of matter in your answer).arrow_forward
- . How many hydrogen atoms are in an alkane that has 7 carbon atoms?arrow_forwardClassify the following organic structures: CH3CCCH3 CH3CONHCH3 And IUPAC name of: CH3-CH2-CH=CH2arrow_forwardAlcoholic beverages contain: a. wood alcohol. b. isopropyl alcohol. c. glyceryl alcohol. d. ethyl alcohol.arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning




