
(a)
Interpretation:
The relationship between the given isomers needs to be established.
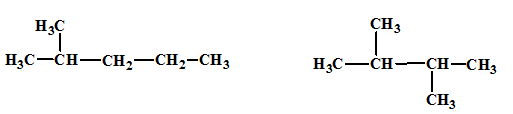
Concept Introduction:
Isomers are two or more compounds which have the same formula but different structures and properties. Constitutional isomers are structural isomers which have the same molecular formula but different connectivity of the constituent atoms.
(b)
Interpretation:
The relationship between the given isomers needs to be established.
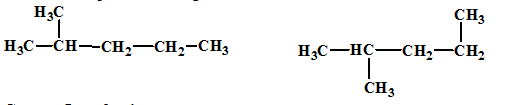
Concept Introduction:
Isomers are two or more compounds which have the same formula but different structures and properties. Constitutional isomers are structural isomers which have the same molecular formula but different connectivity of the constituent atoms.
(c)
Interpretation:
The relationship between the given isomers needs to be established.
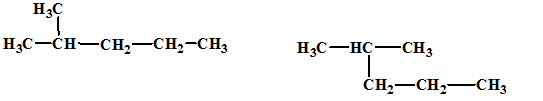
Concept Introduction:
Isomers are two or more compounds which have the same formula but different structures and properties. Constitutional isomers are structural isomers which have the same molecular formula but different connectivity of the constituent atoms.
(d)
Interpretation:
The relationship between the given isomers needs to be established.
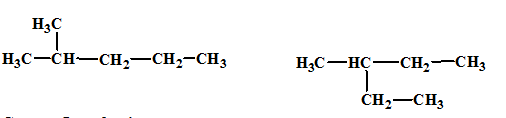
Concept Introduction:
Isomers are two or more compounds which have the same formula but different structures and properties. Constitutional isomers are structural isomers which have the same molecular formula but different connectivity of the constituent atoms.
(e)
Interpretation:
The relationship between the given isomers needs to be established.
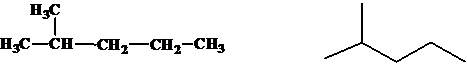
Concept Introduction:
Isomers are two or more compounds which have the same formula but different structures and properties. Constitutional isomers are structural isomers which have the same molecular formula but different connectivity of the constituent atoms.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Why are different conformations of an alkane not considered structural isomers?arrow_forward1a. . Isomers are responsible for the diversity of organic compounds. true or false? b. Positional isomers are molecules with different arrangements of the carbon skeleton but with the same chemical formula. true or false? c.)arrow_forward1. The structures of three compounds are shown in the picture. Why do these substances all belong to the same homologous series? A They all contain an even number of carbon atoms. B They all contain the same functional group. C They are all hydrocarbons. D They are all saturated.arrow_forward
- Complete the following molecular formula of Ethyl acrylate with C, CH, CH2, or CH3.arrow_forwardCyclopentane has a higher boiling point than pentane even though both compounds have the same number of carbons (49 °C vs. 36 °C). Can you suggest a reason for this phenomenon?arrow_forwardHow many structural isomers of C4H6 have exactly one ring?arrow_forward
- 13. When a chain of carbons has all single C-C covalent bonds with a single C=C double bond such that the C backbone looks like this R1-C-C=C-C-R2 this molecule has two very different shapes. These 2 alternate shapes are called: a) geometric isomers b) structural isomers c) stereo isomers d) none are correctarrow_forwardclassify to its functional group CH3-CH2-CH=CH-CH3arrow_forwardDraw the condensed structural formulas and name three constitutional isomers with the formula C4H9Br.arrow_forward
- For each of the following molecules, please name the main functional group present: (a) O (b) O (c) OH ║ ║ │ CH3-C-CH2CH3 CH3-C- OCH3 CH3-CH2-CH2arrow_forwardA. Classify all functional groups B. Classify if they are constitutional isomers, stereoisomers (cis–trans), same or different compoundsarrow_forwardDo isomers exist, if so draw the structures. a. CH3CH2CH=CH2 b. CH3CH2CH=CHCH2CH3arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





