
Concept explainers
(a)
Interpretation:
The
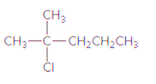
Concept Introduction:
The reactions in which alkane is reacted with halogen, like Cl2 or Br2 is known as halogenation reaction which results in the formation of alkyl halide and hydrogen halide. The important condition for this reaction to occur is heat or exposure to light.
(b)
Interpretation:
The alkane needed as a starting material should be determined which forms following alkyl halides:
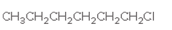
Concept Introduction:
The reactions in which alkane is reacted with halogen, like Cl2 or Br2 is known as halogenation reaction which results in the formation of alkyl halide and hydrogen halide. The important condition for this reaction to occur is heat or exposure to light.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Which of the following reagents would NOT convert the shown alcohol into the alkene a) 1) TsCl, py 2) NaOEt b) NaOEt c) concentrated H2SO4, heatarrow_forward16. How many moles of H2O would be produced by the complete combustion of 1.0 mole of cyclobutane? a. 3.0 b. 4.0 c. 5.0 d. 6.0arrow_forward1. For an addition reaction, why does free energy, ΔG, becomes more positive with increasing temperature? a. The negative enthalpy dominates at high temperature. b. The positive enthalpy dominates at high temperature. c. The positive entropy dominates at high temperature. d. The negative entropy dominates at high temperature 2. Which of the following alkynes will form only 1 product in acid-catalyzed hydration? a. 2-pentyne b. 3-hexyne c. 3-pentyne d. 1-hexyne 3. Which of the following statements best describes the general reactivity of alkynes? a. An alkyne reacts as an electrophile and is, therefore, electron poor. b. Alkynes undergo electrophilic addition reactions just like alkenes. c. An alkyne reacts as an electrophile and is therefore electron-rich. d. An alkyne reacts as a nucleophile and is therefore electron-poor.arrow_forward
- which of the following reagents can oxidize an alkene? H2/Pt, HCl, H2SO4, KMnO4arrow_forwardWhich of the compounds in Figure 5 will react with NaNH₂? * 1- A 2- B 3- C 4- Darrow_forwardWhat alkane is needed as a starting material to prepare each of the following alkyl halides by a halogenation reaction?arrow_forward
- For each of the following alkynes, draw the products obtained from (1) the acid-catalyzed addition of water (mercuric ion is added for part a) and from (2) hydroboration–oxidation: a. 1-butyne b. 2-butyne c. 2-pentynearrow_forward1. How will you carry out the following reactions? a. Combustion b. Bromination c. Oxidation2. Based on their chemical structures (both shape and atomic make-up), do you expect hydrocarbons (in general) to be more, or less, dense than water? Describe how it was done in the lab. 3. What colors are bromine and potassium permanganate solutions? 4. Name a substance/solution that could be used to distinguish alkane from an alkene. 5. Describe the general properties of hydrocarbons based on what you’ve learned in the module.arrow_forwardHow many different alkenes are produced if the following molecule is reacted with sodium ethoxide? A. 1 B. 2 C. 3 D. 4arrow_forward
- 8. What type of reaction takes place when CO2 + H2O and energy are obtained from 3-methyl butyne?a) Hydrogenationb) Halogenationc) Partial oxidationd) Total oxidationarrow_forward46) What is the name of the following: CBr3CH2CCCH3 A. 1,1,1 tribromo, 2-pentyne B. 5,5,5 tribromo, 2-pentene C. 1,1,1 tribromo, 2-pentene D. 1,2,3 tribromo, 2-pentyne E. 5,5,5 tribromo, 2-pentynearrow_forwardWhich of the following alkenes is the most suitable to obtain 5-methyl-4-octanol via hydroboration-oxidation reaction? options: This conversion is not possible through hydroboration. This conversion is only possible through hydration reaction. 4-methyl-3-octene 4-methyl-4-octene 5-methyl-2-octene 5-methyl-3-octenearrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning 
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax





