
Concept explainers
(a)
Interpretation:
The following skeletal structure should be converted to complete structure with all atoms and bond lines.

Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms.
Saturated hydrocarbon is known as
The skeletal structure is also known as line angle formula. The structure of an organic compound is denoted by the shorthand representation of molecule's bonding is known as skeletal structure.
The structure which shows the bond lines between all atoms present in the compound is known as line-bond structure.
(b)
Interpretation:
The following skeletal structure should be converted to complete structure with all atoms and bond lines.
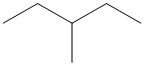
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
Saturated hydrocarbon is known as alkane having general molecular formula
The skeletal structure is also known as line angle formula. The structure of an organic compound is denoted by the shorthand representation of molecule's bonding is known as skeletal structure.
The structure which shows the bond lines between all atoms present in the compound is known as line-bond structure.
(c)
Interpretation:
The following skeletal structure should be converted to complete structure with all atoms and bond lines.
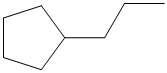
Concept Introduction:
Compounds consist of carbon and hydrogen is known as hydrocarbons. Hydrocarbons are classified as saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that is double and triple bond.
Saturated hydrocarbon is known as alkane having general molecular formula
The skeletal structure is also known as line angle formula. The structure of an organic compound is denoted by the shorthand representation of molecule's bonding is known as skeletal structure.
The structure which shows the bond lines between all atoms present in the compound is known as line-bond structure.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Convert the following condensed formulas into skeletal structures. a. CH3CONHCH3 b. CH3COCH2Br c. (CH3)COH d. CH3COCl e. CH3COCH2CO2H f. HO2CCH(OH)CO2Harrow_forwardConvert the following condensed formulas into skeletal structures. a. CH3CONHCH3b. CH3COCH2Brc. (CH3)3COHd. CH3COCle. CH3COCH2CO2Hf. HO2CCH(OH)CO2Harrow_forwardDefine Skeletal Structures ? explain three rules are used to draw them ?arrow_forward
- Write the condensed and skeletal structures of the given molecule.arrow_forwardDraw the expanded, condensed and skeletal structure for the compound:a. C4H8b. C3H7OHc. C2H5Brd. CH3COOHarrow_forwardplease help with all parts Convert the following condensed structures to Kekule´Kekule´ structures, showing the covalent bonds. Draw the molecule by placing atoms on the canvas and connecting them with bonds. Include all hydrogen atoms and nonbonding electrons.: part 1) (CH3)2CHCl part2) (CH3)3CBr part 3) (CH3)3C(CH2)3CHOarrow_forward
- Write condensed structural formulas, and skeletal structures for each structural isomer of C4H10.arrow_forwardConvert each condensed formula to a Lewis structure. a.CH3(CH2)4CH(CH3)2 b. (CH3)3CCH(OH)CH2CH3 c. (CH3)2CHCHO d.(HOCH2)2CH(CH2)3C(CH3)2CH2CH3arrow_forwardWhat is the condensed structure for the lewis structure and the skeletal structure?arrow_forward
- draw skeletal structure of this molecule: H2N-CH2-CH-CH2-CH3-CH3arrow_forwardHow many electron pairs are shared when a triple bond exists between two carbon atoms? What must he the geometric arrangement around the carbon atoms in a triple bond? Draw the Lewis structure of a simple molecule that contains a triple bond.arrow_forwardDraw a skeletal structure for each condensed structure given and draw a condensed structure for each skeletal structure.arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
