
Concept explainers
Interpretation:
Two sets of reagents that might be combined in a Wittig reaction to give 1-Phenyl-1,3-pentadiene has to be suggested.
Concept introduction:
The functional group in the
An
Compounds with two double bonds are known as dienes.
Wittig reaction is a reaction in which a nucleophilic elimination occurs after nucleophilic addition reaction. In this reaction an aldehyde or ketone reacts with a triphenyl phosphonium ylide (Wittig reagent) to give an alkene and triphenylphosphine oxide.
The reaction can be represented as shown below,
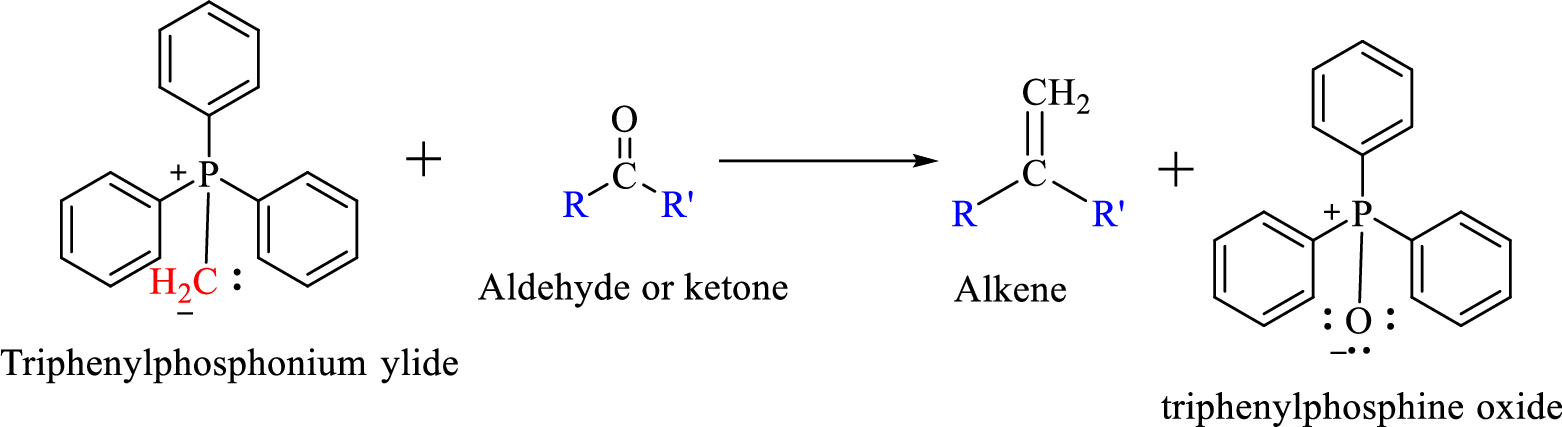
Mechanism:
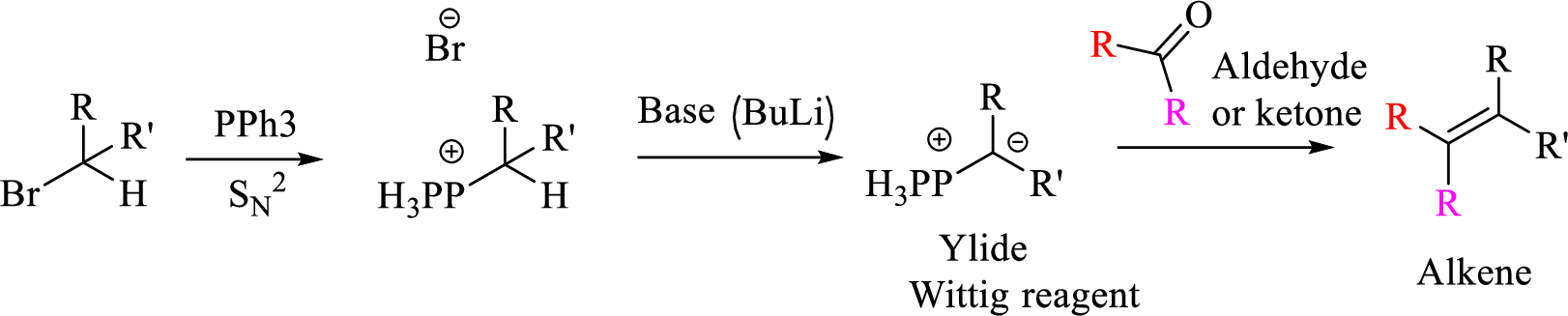
Trending nowThis is a popular solution!

Chapter 16 Solutions
Organic Chemistry
- Following is a retrosynthetic scheme for the synthesis of the tricyclic diene on the left. Show how to accomplish this synthesis from 2-bromopropane, cyclopentadiene, and 2-cyclohexenone.arrow_forwardBicyclo-2,5-heptadiene can be prepared in two steps from cyclopentadiene and vinyl chloride. Provide a mechanism for each step.arrow_forwardOne step in the synthesis of dodecahedrane involves reaction of the tetraene C with dimethylacetylene dicarboxylate (D) to afford two compounds having molecular formula C16H16O4. This reaction has been called a domino Diels–Alder reaction. Identify the two products formed.arrow_forward
- Provide the products of the reaction of 1-propene with HBr in the presence and absence of peroxide. Explain the mechanism of the formation of the products in detailarrow_forwardAddition of HBr to 3,3-dimethyl-1-butene gives a mixture of two isomeric alkyl bromide products. Draw structures for the two products, and give a mechanistic explanation for their formation.arrow_forwardWhen anthracene is added to the reaction of chlorobenzene with concentrated NaOH at 350 °C, an interesting Diels–Alderadduct of formula C20H14 results. The proton NMR spectrum of the product shows a singlet of area 2 around d 3 and abroad singlet of area 12 around d 7. Propose a structure for the product, and explain why one of the aromatic rings ofanthracene reacted as a dienearrow_forward
- Addition of HBr to 3,3-dimethyl-1-butene gives a mixture of two isomeric alkyl bromide products. Please daw structures for the two products, and give a mechanistic explanation for their formation.arrow_forwardBased on 1,3-cyclopentadiene, using the necessary organic and inorganic chemicals. The synthesis scheme includes the synthesis of the following compound, including the Diel-Alder reaction design.arrow_forwardIn an electrophilic addition reaction, conjugated dienes give more than one expected product. Show the products when using the reagent 1,3 –butadiene and HBr.arrow_forward
- When 1 mole of buta-1,3-diene reacts with 1 mole of HBr, both 3-bromobut-1-ene and 1-bromobut-2-ene are formed. Propose a mechanism to account for this mixture of products.arrow_forwardPropose two sets of reagents that might be combined in a Wittig reaction to give the indicated Z alkenearrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


