
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16.8, Problem BQ
The given mechanism of transamination reaction is shown below,
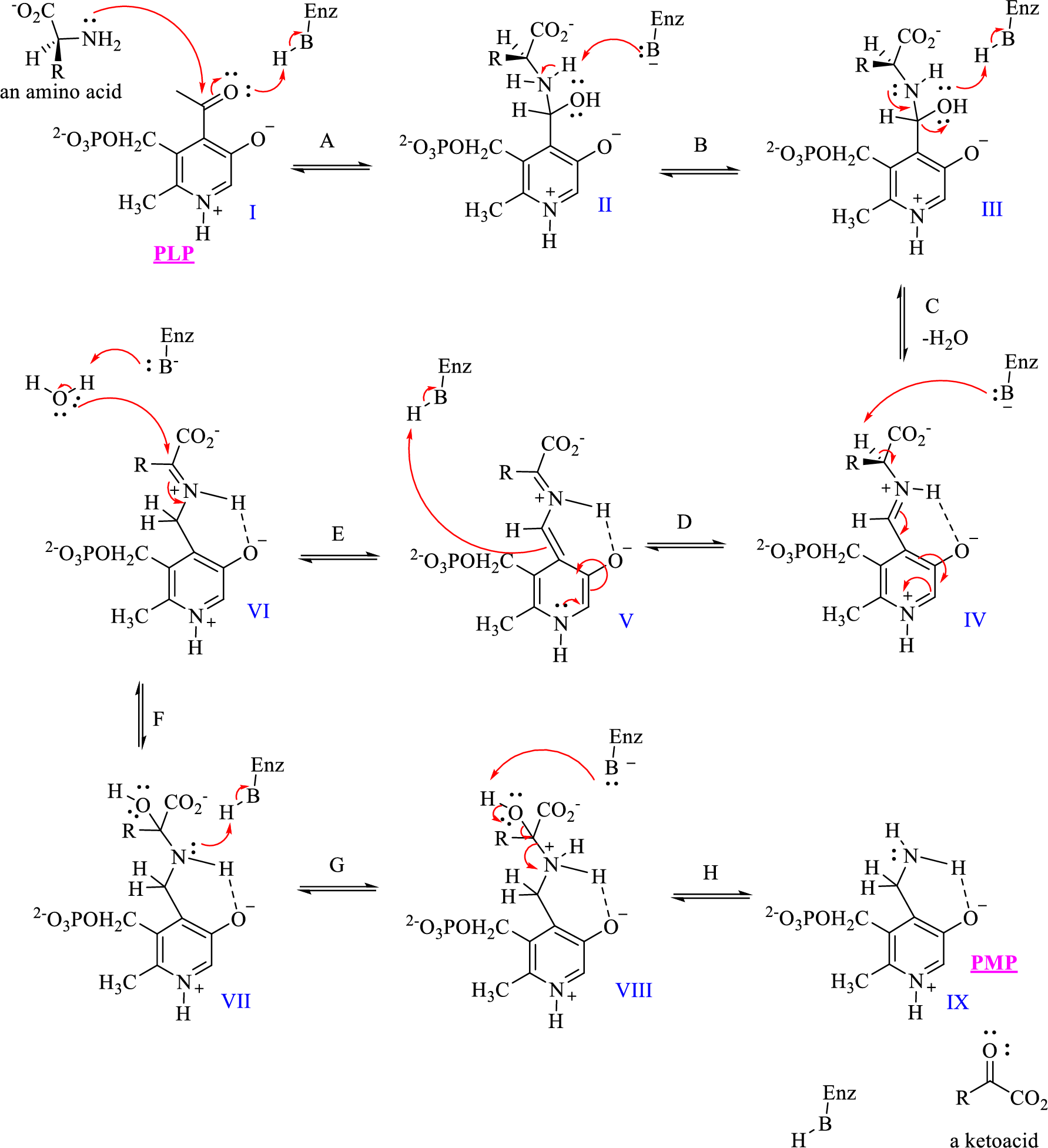
Which structures would be classified as tetrahedral intermediates derived from addition reactions?
- 1. II, III, VII, and VIII
- 2. IV, V, and VI
- 3. II, IV, and VIII
- 4. II and VI
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which of the following reaction is mot likely to activate a drug molecule from orally administered inactive parent molecule?
a
Aromatic hydroxylation
b
Benzylic carbon oxidation
c
Hydrolysis
d
Azo reduction
PRODUCTS. Match the name of the product with the biochemical reaction. Products may be used more than once. Tests may require more than one answer.a. 2,3-butanediol 1.catalase b. ammonia 2.phenylalanine deamination c.fatty acids 3.triglyceride hydrolysis d. indole 4.tryptophan degradation e.molecular oxygen 5.urea hydrolysis f. phenylpyruvic acid 6.Voges-Proskauer test
As shown in Mechanism 29.2, the nal steps in the Edman degradation result in rearrangement of a thiazolinone to an Nphenylthiohydantoin. Draw a stepwise mechanism for this acid-catalyzed reaction.Mechanism 29.2: Edman Degradation
Chapter 16 Solutions
Organic Chemistry
Ch. 16.1 - Write the IUPAC name for each compound. Specify...Ch. 16.1 - Write structural formulas for all aldehydes with...Ch. 16.1 - Write the IUPAC name for each compound.Ch. 16.5 - Prob. 16.4PCh. 16.6 - Prob. 16.5PCh. 16.7 - Prob. 16.6PCh. 16.7 - Write a mechanism for the acid-catalyzed...Ch. 16.8 - Prob. 16.8PCh. 16.8 - The given mechanism of transamination reaction is...Ch. 16.8 - The given mechanism of transamination reaction is...
Ch. 16.8 - The given mechanism of transamination reaction is...Ch. 16.8 - Prob. DQCh. 16.8 - Prob. EQCh. 16.8 - The given mechanism of transamination reaction is...Ch. 16.9 - Predict the position of the following equilibrium.Ch. 16.9 - Draw a structural formula for the keto form of...Ch. 16.10 - Prob. 16.11PCh. 16.11 - What aldehyde or ketone gives these alcohols upon...Ch. 16.11 - Prob. 16.13PCh. 16 - Prob. 16.14PCh. 16 - Prob. 16.15PCh. 16 - The infrared spectrum of compound A, C6H12O, shows...Ch. 16 - Following are 1H-NMR spectra for compounds B...Ch. 16 - Draw structural formulas for the product formed by...Ch. 16 - Suggest a synthesis for the following alcohols...Ch. 16 - Show how to synthesize the following alcohol using...Ch. 16 - 1-Phenyl-2-butanol is used in perfumery. Show how...Ch. 16 - Prob. 16.22PCh. 16 - Draw structural formulas for (1) the...Ch. 16 - Show how to bring about the following conversions...Ch. 16 - Prob. 16.25PCh. 16 - Wittig reactions with the following -chloroethers...Ch. 16 - Prob. 16.27PCh. 16 - Prob. 16.28PCh. 16 - 5-Hydroxyhexanal forms a six-membered cyclic...Ch. 16 - Prob. 16.30PCh. 16 - Prob. 16.31PCh. 16 - Propose a mechanism to account for the formation...Ch. 16 - Prob. 16.33PCh. 16 - Prob. 16.34PCh. 16 - Show how to bring about the following conversion.Ch. 16 - A primary or secondary alcohol can be protected by...Ch. 16 - Prob. 16.37PCh. 16 - Prob. 16.38PCh. 16 - Prob. 16.39PCh. 16 - Prob. 16.40PCh. 16 - The following molecule belongs to a class of...Ch. 16 - When cis-2-decalone is dissolved in ether...Ch. 16 - Prob. 16.43PCh. 16 - Prob. 16.44PCh. 16 - The following bicyclic ketone has two -carbons and...Ch. 16 - Propose a mechanism for this reaction.Ch. 16 - The base-promoted rearrangement of an -haloketone...Ch. 16 - If the Favorskii rearrangement of...Ch. 16 - (R)-Pulegone, readily available from pennyroyal...Ch. 16 - (R)-Pulegone is converted to (R)-citronellic acid...Ch. 16 - Starting with cyclohexanone, show how to prepare...Ch. 16 - Show how to convert cyclopentanone to these...Ch. 16 - Prob. 16.53PCh. 16 - Prob. 16.54PCh. 16 - Prob. 16.55PCh. 16 - Following is the structural formula of Surfynol, a...Ch. 16 - Prob. 16.57PCh. 16 - Propose a mechanism for this isomerization.Ch. 16 - Starting with acetylene and 1-bromobutane as the...Ch. 16 - Prob. 16.60PCh. 16 - Prob. 16.61PCh. 16 - Prob. 16.62PCh. 16 - Prob. 16.63PCh. 16 - Prob. 16.64PCh. 16 - All rearrangements we have discussed so far have...Ch. 16 - In dilute aqueous base, (R)-glyceraldehyde is...Ch. 16 - Treatment of -D-glucose with methanol in the...Ch. 16 - Treating a Grignard reagent with carbon dioxide...Ch. 16 - Prob. 16.69PCh. 16 - Prob. 16.70PCh. 16 - Prob. 16.71PCh. 16 - Prob. 16.72PCh. 16 - Write the products of the following sequences of...Ch. 16 - Using your reaction roadmaps as a guide, show how...Ch. 16 - Using your reaction roadmaps as a guide, show how...Ch. 16 - Using your reaction roadmaps as a guide, show how...Ch. 16 - Using your reaction roadmaps as a guide, show how...Ch. 16 - Prob. 16.78PCh. 16 - Prob. 16.79PCh. 16 - Prob. 16.80PCh. 16 - Prob. 16.81P
Additional Science Textbook Solutions
Find more solutions based on key concepts
Determine the de Brogue wavelength of a. an electron moving at 1/10 the speed of light. b. a 400 g Frisbee movi...
Inorganic Chemistry
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
Give one example from main group chemistry that illustrates each of the following descriptions: (a) Covalent ne...
General Chemistry: Atoms First
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (14th Edition)
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (6th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The given mechanism of transamination reaction is shown below, Which structures are imine or iminium intermediates? 1. I, III, and V 2. VII and VIII 3. IV and VII 4. IV, V, and VIarrow_forwardThe given mechanism of transamination reaction is shown below, What is the role of the pyridinium group in Step D? 1. It acts as an electron-donating group (source) of electrons to enhance deprotonation of the amino acid by the enzyme. 2. It acts as an electron-withdrawing group (sink) to facilitate deprotonation of the amino acid. 3. It plays the role of a Lewis base to coordinate the amino acid. 4. Both 2 and 3 are true.arrow_forwardOne of the steps in the metabolic degradation of guanine is hydrolysis to give xanthine. Propose a mechanism.arrow_forward
- The given mechanism of transamination reaction is shown below, To complete the shuffling of amino groups and carbonyls, the PMP must be converted back to PLP. How would you predict that this occurs? 1. A separate reaction oxidizes the amine group of PMP to an aldehyde. 2. A separate reaction hydrolyzes the amine of PMP to an aldehyde. 3. A different ketoacid reacts with the amine of PMP, and the entire sequence runs backward. 4. Either 2 or 3 could occur.arrow_forward1. Biogenic Amines are inactivated primarily by a. Sulfonation b. Glucoronidation c. Aromatic hydroxylation d. Methylation 2. A drug can exert its pharmacological effect only if it is a. Protein bound b. Protein unbound c. Free drug d. Both B & C e. Both A & C 3. In order for the drug to be ready and available for absorption, it must be release first from its dosage form with the exception of: a. Capsule b. Tablet c. Solution d. Suspension 4. All of the following are true, except a. Solubility increase with decrease particle size b. Solubility increase with increase surface area c. Solubility increase with increase particle size d. Solubility decrease with decrease surface area 5. The rate in which the drug appears in the bloodstream is also known as a. Half-life b. Potency c. Bioavailability d. Area under the curvearrow_forwardWhat is the mechanism of action of sarin or a sarin like chemical? Group of answer choices It binds to acetylcholinesterase (AChE) and makes it work faster It binds to Acetylcholine (ACh) and decreases its affinity for cholinergic receptors It binds to Acetylcholine (ACh) and increases its affinity for cholinergic receptors It binds to acetylcholinesterase (AChE) and makes it unable to function pick one answerarrow_forward
- 4(a) Suggest a test you will use to show that a given food substance contains protein.(b)Show how you will use(i)Modified Gabriel’s Synthsis(ii)Streckers’s Synthesis to prepare phenylalanine in the laboratory.arrow_forward4) Draw and note the similarities of the cyanohydrin anion to Vitamin B1 ylide and BMIMCl ylide analyzing why pKa is in similar range 5) Analogously to the preparative stage of the citric acid cycle, pyruvic acid undergoes an oxidative decarboxylation to form acetaldehyde using the VitB1 ylide anion. Draw out this mechanism Please answer very soon will give rating surely Both questions answers neededarrow_forwardplease please help me to answer it >>> i have an exam Human xanthine oxidase catalyzes the oxidation of hypoxanthine to xanthine and can furthercatalyze the oxidation of xanthine to uric acid. For the treatment of hyperuricemia and gout,several medications are used to inhibit the activity of xanthine oxidase and reduce theproduction of uric acid. You are a biochemist and just discovered a chemical that can inhibitthe activity of the human xanthine oxidase. When analyzing its mode of inhibition, you foundthat the enzyme inhibitor complex requires 450 J.mol-1 to dissociate and that it displayskinetics somehow similar to noncompetitive inhibition. You sent your inhibitor to the ministryof health for approval as a medication for gout. Based on the data provided, are they going toauthorize it as a medication or not? Explain?arrow_forward
- Bacteria that live in the large intestine metabolize glycocholic acid using amidases and hydroxylases. (a) Draw the reaction product. (b) Compare the solubility of the product and glycocholic acid. (c) Could the change in the bile acid’s solubility make it less toxic to bacteria?arrow_forwardCells can also convert a-keto acids into amino acids, but because the reagents organic chemists use for this reaction are not available in cells, they carry out this reaction by a different mechanism. a. What amino acid is obtained from the reductive amination of each of the following metabolic intermediates in a cell by reductive amination? b. What amino acids are obtained from the same metabolic intermediates when the amino acids are synthesized in the laboratory?arrow_forwardMatch the following drug metabolism pathway Choices: 1.Acetylsalicylic acid Hydrolysis 2. Methylphenidate Oxidation 3. Clonazepam Reduction 4. Chloroform 5. Procainamidearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning


DIGESTER-35 | VITAMINS AND THEIR RELATED COENZYMES| GPAT | NIPER | PHARMACIST| DI; Author: GPAT DISCUSSION CENTER;https://www.youtube.com/watch?v=CGrdNYmho0s;License: Standard YouTube License, CC-BY