
Concept explainers
Interpretation: The nucleophilic addition reaction occuring steps in the given transamination reaction has to be determined.
Concept introduction:
Amino acids are the molecules containing an
Carbon, hydrogen, oxygen and nitrogen are the key elements in amino acid.
General structure of an amino acid can be drawn as follows,
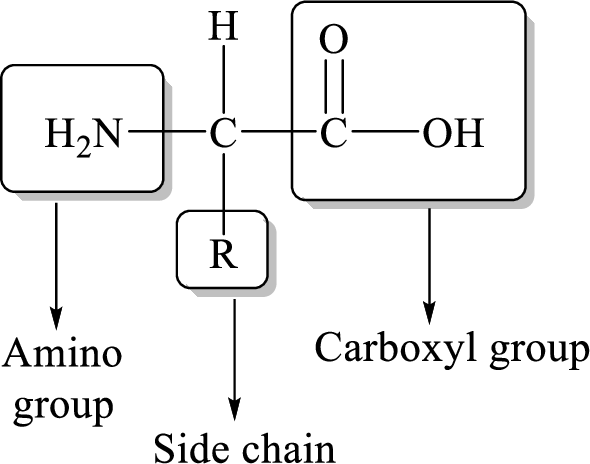
The transfer of an amino group from an amino acid to a ketoacid with the formation of a new amino acid and a new keto acid is known as transamination reaction.
This reaction is catalysed by a group of enzymes called transaminases or aminotransferases. PLP- Pyridoxalphosphate acts as co-factor in this reaction.
Nucleophile: It is negatively charged species which seeks for positive charge and hence donate pair of electrons to positively charged species (electrophiles) which results in the formation of
Nucleophilic addition reaction is an addition reaction where in a chemical compound a
In a nucleophilic addition reaction the first step is the bond formation between a nucleophile and electrophile.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Organic Chemistry
- The given mechanism of transamination reaction is shown below, Which structures are imine or iminium intermediates? 1. I, III, and V 2. VII and VIII 3. IV and VII 4. IV, V, and VIarrow_forwardThe given mechanism of transamination reaction is shown below, Which structures would be classified as tetrahedral intermediates derived from addition reactions? 1. II, III, VII, and VIII 2. IV, V, and VI 3. II, IV, and VIII 4. II and VIarrow_forwardPeppermint essential oil has been a classic choice for the treatment of ________ for hundreds of years. Choose one answer. a. moles b. incontinence c. measles d. nauseaarrow_forward
- Match the following drug metabolism pathway Choices: 1.Acetylsalicylic acid Hydrolysis 2. Methylphenidate Oxidation 3. Clonazepam Reduction 4. Chloroform 5. Procainamidearrow_forwardCells can also convert a-keto acids into amino acids, but because the reagents organic chemists use for this reaction are not available in cells, they carry out this reaction by a different mechanism. a. What amino acid is obtained from the reductive amination of each of the following metabolic intermediates in a cell by reductive amination? b. What amino acids are obtained from the same metabolic intermediates when the amino acids are synthesized in the laboratory?arrow_forwardThe given mechanism of transamination reaction is shown below, What is the role of the pyridinium group in Step D? 1. It acts as an electron-donating group (source) of electrons to enhance deprotonation of the amino acid by the enzyme. 2. It acts as an electron-withdrawing group (sink) to facilitate deprotonation of the amino acid. 3. It plays the role of a Lewis base to coordinate the amino acid. 4. Both 2 and 3 are true.arrow_forward
- One of these reactions is a reductive amination. Write the oxidation states ( a number) next to each boxed carbon. Which reaction is a reductive amination? A or B Identify the oxidized and reduced states of the coenzymesarrow_forwardChoose a different family of natural products derived from the acetate pathway and propose how such pathway would overlap with the cannabinoid?arrow_forwarda) draw the structure of the tetrahedral intermediate INITIALLT-FORMED in the reaction shown. b) Draw the structures of the organic products of the acyl transfer reaction.arrow_forward
- ① H+ + HCO3─ + Pi → Carboxyphosphate + H2O ∆G0’ =+19.7 kJ mol-1 ② ATP + H2O → ADP + Pi + H+ ∆G0’ = ─30.5 kJ mol-1 ③ Carboxyphosphate + pyruvate → oxaloacetate + Pi ∆G0’ = ??? kJ mol-1 ④ Overall reaction: ATP + HCO3─ + pyruvate → oxaloacetate + ADP + Pi ∆G0’ = ─2.1 kJ mol-1 a) Use reactions ①, ② and overall reaction ④ to calculate the ∆G0’ for the formation of oxaloacetate from pyruvate and carboxyphosphate (reaction ③ above). Use reactions ①, ② and overall reaction ④ to calculate the ∆G0’ for the formation of oxaloacetate from pyruvate and carboxyphosphate (reaction ③ above). b) Now calculate ∆G0’ for the formation of oxaloacetate from pyruvate and bicarbonate in the absence of ATP. c) Explain the difference in your answers to parts a and b.arrow_forwardWhy can't insulin be taken orally? It does not taste very good Enzymes in saliva break it down Hydrolysis of insulin occurs in the stomach It is not possible to store insulinarrow_forwardplease please help me to answer it >>> i have an exam Human xanthine oxidase catalyzes the oxidation of hypoxanthine to xanthine and can furthercatalyze the oxidation of xanthine to uric acid. For the treatment of hyperuricemia and gout,several medications are used to inhibit the activity of xanthine oxidase and reduce theproduction of uric acid. You are a biochemist and just discovered a chemical that can inhibitthe activity of the human xanthine oxidase. When analyzing its mode of inhibition, you foundthat the enzyme inhibitor complex requires 450 J.mol-1 to dissociate and that it displayskinetics somehow similar to noncompetitive inhibition. You sent your inhibitor to the ministryof health for approval as a medication for gout. Based on the data provided, are they going toauthorize it as a medication or not? Explain?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning




