
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
9th Edition
ISBN: 9780136781776
Author: Wade
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 16.33SP
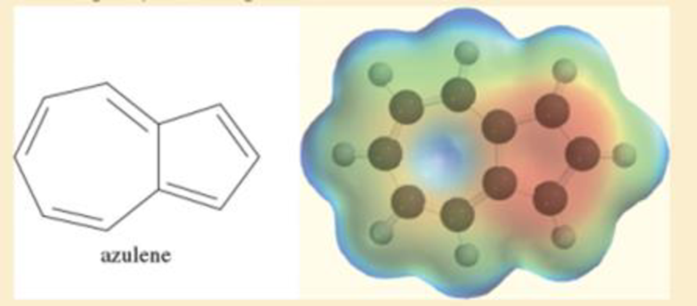
Azulene is a deep-blue hydrocarbon with resonance energy of 205 kJ/mol (49 kcal/mol). Azulene has ten pi electrons, so it might be considered as one large
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Azulene, an isomer of naphthalene, has a remarkably large dipole moment for a
hydrocarbon (µ = 1.0 D). Explain using resonance structures.
Azulene is a deep-blue hydrocarbon with resonance energy of 205 kJ>mol (49 kcal>mol). Azulene has ten pi electrons, so it might be considered as one large aromatic ring. Its electrostatic potential map shows one ring to be highly electron-rich (red) and the other to be electron-poor (blue). The dipole moment is unusually large (1.0 D) for a hydrocarbon. Show how this charge separation might arise.
Acridine is a heterocyclic aromatic compound obtained from coal tar that is used in the synthesis of dyes. The molecular formula of acridine is C13H9N, and its ring system is analogous to that of anthracene except that one CH group has been replaced by N. The two most stable resonance structures of acridine are equivalent to each other, and both contain a pyridine-like structural unit. Write a structural formula for acridine.
Chapter 16 Solutions
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
Ch. 16.2 - Prob. 16.1PCh. 16.2 - Prob. 16.2PCh. 16.2 - a. Draw the resonance forms of benzene,...Ch. 16.2 - Show the product of the Diels-Alder dimerization...Ch. 16.4 - Prob. 16.5PCh. 16.6 - Make a model of cyclooctatetraene in the tub...Ch. 16.6 - Prob. 16.7PCh. 16.6 - Prob. 16.8PCh. 16.7 - Prob. 16.9PCh. 16.8A - a. Draw the molecular orbitals for the...
Ch. 16.8A - Repeat Problem16-10 for the cyclopentadienyl ions....Ch. 16.8C - Explain why each compound or ion should be...Ch. 16.8C - The following hydrocarbon has an unusually large...Ch. 16.8C - Prob. 16.14PCh. 16.8C - Prob. 16.15PCh. 16.9B - Prob. 16.16PCh. 16.9C - Show which of the nitrogen atoms in purine are...Ch. 16.9C - The proton NMR spectrum of 2-pyridone gives the...Ch. 16.9D - Prob. 16.19PCh. 16.9D - Prob. 16.20PCh. 16.10 - Prob. 16.21PCh. 16.12 - Ciprofloxacin is a member of the fluoroquinolone...Ch. 16.13 - Draw and name all the chlorinated benzenes having...Ch. 16.13 - Name the following compounds:Ch. 16.15 - The UV spectrum of 1-phenylprop-2-en-1-ol shows an...Ch. 16 - Prob. 16.26SPCh. 16 - Name the following compounds:Ch. 16 - Draw and name all the methyl, dimethyl, and...Ch. 16 - Four pairs of compounds are shown. In each pair,...Ch. 16 - One of the following hydrocarbons is much more...Ch. 16 - In Kekuls time cyclohexane was unknown, and there...Ch. 16 - Prob. 16.32SPCh. 16 - Azulene is a deep-blue hydrocarbon with resonance...Ch. 16 - Prob. 16.34SPCh. 16 - Prob. 16.35SPCh. 16 - Prob. 16.36SPCh. 16 - Prob. 16.37SPCh. 16 - Prob. 16.38SPCh. 16 - Prob. 16.39SPCh. 16 - Biphenyl has the following structure. a. Is...Ch. 16 - Anions of hydrocarbons are rare, and dianions of...Ch. 16 - How would you convert the following compounds to...Ch. 16 - Prob. 16.43SPCh. 16 - Prob. 16.44SPCh. 16 - A student found an old bottle labeled thymol on...Ch. 16 - Prob. 16.46SPCh. 16 - Prob. 16.47SPCh. 16 - Prob. 16.48SPCh. 16 - The proton NMR chemical shifts of the hydrogens in...Ch. 16 - Prob. 16.50SPCh. 16 - NMR has been used to probe many molecular...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The compound shown reacts readily with HBr and adds water across at least of one of its double bonds. Why doesn't it behave like Benzene? 1. It is NOT a 4n + 2 system 2. severe steric interactions prevent it from being flat 3. It does NOT have a continous array of p orbitals to overlap 4. There are too many pi bonds to be aromaticarrow_forwardFor the following molecules, show how the aryl substituents impact the electron density at the unique carbons around the aromatic ring by showing all unique resonance structures, including appropriate formal charges. OMe Me MeO Mearrow_forwardFollowing the instructions for drawing the energy levels of the molecular orbitals for the compounds shown in Figure 8.17, draw the energy levels of the molecular orbitals for the cycloheptatrienyl cation, the cycloheptatrienyl anion, and the cyclopropenyl cation. For each compound, show the distribution of the p electrons. Which of the compounds are aromatic?arrow_forward
- (b) The structure of heptalene (2E) is shown below. Answer the following questions: (i) Is heptalene aromatic, non-aromatic, or anti-aromatic? (ii) 2E can be readily protonated to form a more stable species. Draw the structure of the protonated 2E and explain the stability. heptalene (2E)arrow_forwardAromatic heterocyclic chemistry: State whether the following drugs are aromatic heterocycles and if they follow the 4 aromaticity rules (unsaturation, (4n + 2)pi electrons, cyclic and flat/planar), NAME ANY HETEROCYCLES. Draw the structure of the core of any aromatic heterocycles in the drug. Riociguat, Imatinib, Clopidogrel, Glatiramer acetate, Axitinibarrow_forward1. There are several isomeric alkanes of molecular formula C6H14.Two of these exhibit the following 1H-NMR spectra. Propose a structure for each of the isomers. Isomer A: δ = 0.84 (d, 12 H), 1.39 (septet, 2H) ppm Isomer B: δ = 0.84 (t, 3 H), 0.86 (s, 9H), 1.22 (q, 2H) ppmarrow_forward
- 4 – Use bond-dissociation enthalpies (pg 167 of text) to calculate the DHo for each of the following reactions. CH3CH2CH3 + H2 g CH3CH3 + CH4 CH3CH2Cl + HI g CH3CH2I + HClarrow_forwardWhich of the following compounds are aromatic and anti-aromatic? Give justifications for your answer. i) ii) iii) +, iv)arrow_forwardPlease rank the below aromatics in order of increasing electron richness (least to most). OMe ܘ ܘ ܘ 2 NH₂ Br 3 4 5 OMe 6arrow_forward
- 1. Guanine (1) is commonly represented as a non aromatic, partially saturated heterocycle. However, the common structure of guanine is only one of two possible resonance forms, of whom the second one is clearly aromatic. Knowing that the latter is originated by the delocalisation of one of N6 (nitrogen six) lone pair on the ring, would you be able to predict the structure of the aromatic resonance structure of guanine? Guanine 6 N. 'NH 3 R 1 Why cannot N4 delocalise its lone pair on the ring? What about N3? Is its lone pair delocalised on the pyrimido-imidazole ring? Why?arrow_forwardGive reasons for the following :(i) Phenol is more acidic than methanol.(ii) The C—O—H bond angle in alcohols is slightly less than the tetrahedral angle (190°28′).(iii) (CH3)3C—O—CH3 on reaction with HI gives (CH3)3C—I and CH3—OH as the main products and not (CH3)3C—OH and CH3—I.arrow_forwardThe C‚N triple bond in acetonitrile has a dipole moment of about 3.6 D and a bond length of about 1.16 Å. Calculate the amount of charge separation in this bond. How important is the charge-separated resonance form in the structure of acetonitrile?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY