
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
9th Edition
ISBN: 9780136781776
Author: Wade
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 16.51SP
NMR has been used to probe many molecular properties, including aromaticity. One of the interesting electronic effects of
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
(b) One isomer of dimethoxybenzoic acid has 1H NMR spectrum dµ (ppm) 3.85 (6H, s), 6.63 (1H, t, J 2 Hz),
and 7.17 (2H, d, J 2Hz), One isomer of coumalic acid has 'H NMR spectrum dн (ppm) 6.41 (1H, d, J 10
Hz), 7.82 (1H, dd, J 2 Hz, 10 Hz) and 8.51 (1H, d, J 2Hz). In each case, which isomer is represented
here? The bonds sticking into the centre of the ring can be to any carbon atom.
Note: COOH proton is not indicated in the spectra
MeO
HO₂C
CO2H
MeO
dimethoxybenzoic acid
coumalic acid
Following are 'H-NMR and BC-NMR spectral data for compound G (CH0). From
this information, deduce the structure of compound G.
'H-NMR
13C-NMR
2.50 (t, 2H)
210.19 126.82
3.05 (t, 2H)
136.64 126.75
3.58 (s, 2H)
133.25
45.02
7.1-7.3 (m, 4H)
128.14
38.11
127.75
28.34
Draw a molecular orbital diagram for the pi system of tetrazole (including the piacemen oi iis pi electrons) and provide
an analysis of its aromaticity using Hückel's rule denoting whether tetrazole is in fact aromatic, antiaromatic, or nonaromatic.
H
N°
Tetrazole
b. Provide a picture of the Highest occupied Molecular Orbital (HOMO) and the Lowest Unnocupied Molecular Orbital
(LUMO) of tetrazole depicting the phases of thep orbitals, the bonding, antibonding, and non-bonding interactions.
Chapter 16 Solutions
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
Ch. 16.2 - Prob. 16.1PCh. 16.2 - Prob. 16.2PCh. 16.2 - a. Draw the resonance forms of benzene,...Ch. 16.2 - Show the product of the Diels-Alder dimerization...Ch. 16.4 - Prob. 16.5PCh. 16.6 - Make a model of cyclooctatetraene in the tub...Ch. 16.6 - Prob. 16.7PCh. 16.6 - Prob. 16.8PCh. 16.7 - Prob. 16.9PCh. 16.8A - a. Draw the molecular orbitals for the...
Ch. 16.8A - Repeat Problem16-10 for the cyclopentadienyl ions....Ch. 16.8C - Explain why each compound or ion should be...Ch. 16.8C - The following hydrocarbon has an unusually large...Ch. 16.8C - Prob. 16.14PCh. 16.8C - Prob. 16.15PCh. 16.9B - Prob. 16.16PCh. 16.9C - Show which of the nitrogen atoms in purine are...Ch. 16.9C - The proton NMR spectrum of 2-pyridone gives the...Ch. 16.9D - Prob. 16.19PCh. 16.9D - Prob. 16.20PCh. 16.10 - Prob. 16.21PCh. 16.12 - Ciprofloxacin is a member of the fluoroquinolone...Ch. 16.13 - Draw and name all the chlorinated benzenes having...Ch. 16.13 - Name the following compounds:Ch. 16.15 - The UV spectrum of 1-phenylprop-2-en-1-ol shows an...Ch. 16 - Prob. 16.26SPCh. 16 - Name the following compounds:Ch. 16 - Draw and name all the methyl, dimethyl, and...Ch. 16 - Four pairs of compounds are shown. In each pair,...Ch. 16 - One of the following hydrocarbons is much more...Ch. 16 - In Kekuls time cyclohexane was unknown, and there...Ch. 16 - Prob. 16.32SPCh. 16 - Azulene is a deep-blue hydrocarbon with resonance...Ch. 16 - Prob. 16.34SPCh. 16 - Prob. 16.35SPCh. 16 - Prob. 16.36SPCh. 16 - Prob. 16.37SPCh. 16 - Prob. 16.38SPCh. 16 - Prob. 16.39SPCh. 16 - Biphenyl has the following structure. a. Is...Ch. 16 - Anions of hydrocarbons are rare, and dianions of...Ch. 16 - How would you convert the following compounds to...Ch. 16 - Prob. 16.43SPCh. 16 - Prob. 16.44SPCh. 16 - A student found an old bottle labeled thymol on...Ch. 16 - Prob. 16.46SPCh. 16 - Prob. 16.47SPCh. 16 - Prob. 16.48SPCh. 16 - The proton NMR chemical shifts of the hydrogens in...Ch. 16 - Prob. 16.50SPCh. 16 - NMR has been used to probe many molecular...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
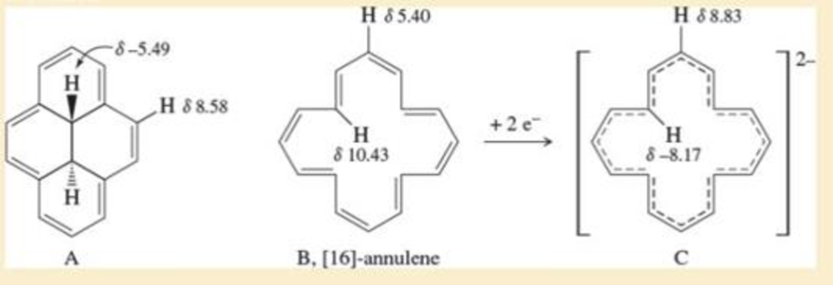
- Propose a structural formula for each compound consistent with its 1H-NMR and 13C-NMR spectra.arrow_forwardGiven here are 1H-NMR and 13C-NMR spectral data for two compounds. Each shows strong, sharp absorption between 1700 and 1720 cm-1 and strong, broad absorption over the region 2500–3000 cm-1. Propose a structural formula for compound.arrow_forwardUse the observed 1H NMR data to decide whether C and its dianion are aromatic, antiaromatic, or not aromatic. C shows NMR signals at –4.25 (6 H) and 8.14–8.67 (10 H) ppm. The dianion of C shows NMR signals at –3 (10 H) and 21 (6 H) ppm. Why are the signals shifted upeld (or downeld) to such a large extent?arrow_forward
- Construct a structure of the given 1H NMR data signals.A. C5H12O 0.91 δ (3H, triplet) 1.19 δ (6H, singlet) 1.50 δ (2H, quartet) 2.24 δ (1H, singlet) B. C4H10O 0.90 δ (6H, doublet) 1.76 δ (1H, multiplet) 3.38 δ (2H, doublet) 3.92 δ (1H, singlet) C. C5H10O 1.09 δ (6H, doublet) 2.12 δ (3H, singlet) 2.58 δ (1H, septet)arrow_forwardThere is a Compound called A with the formula C13H20N2O2. In the proton NMR two triplets at 2.8 and 4.3 are coupled to each other. Similarly, a triplet at 1.1 and a quartet at 2.6 are coupled to each other. the carbonyl group appearing at 1669 cm -1 in the IR spectrum has an unusually low value. Please provide the structure of A.arrow_forwardstep by step show details how to do this question?I am confusedarrow_forward
- Compound A has molecular formula C5H8Br4 but shows only one singlet in the 1H-NMR spectrum. Suggest a structure for A and explain your reasoning.arrow_forward1. Propose the structure of a compound that exhibits the following 'H NMR data: a. CsH10O 1.09 8 (6H, doublet) 2.128 (3H, singlet) 2.588 (1H, septet) b. CH2O 0.91 8 (3H, triplet) 1.198 (6H, singlet) 1.50 8 (2H, quartet) 2.24 ō (1H, singlet)arrow_forwardCc.18.arrow_forward
- 6. Determine the structures of the following aromatic compounds based on their NMR spectra. Copyright McGraw-Hill Education. All rights reserved. No reproduction or distribution without the prior written consent of McGraw-Hill Education. a. C,0H14: IR absorptions at 3150-2850, 1600, and 1500 cm b. CoH12: C NMR signals at 21, 127, and 138 ppm H NMR spectrum 6 H H NMR spectrum 9 H 3 H 1 H 3 H 4 H 8. 6. 5. 4 2 6. 4 ppm ppm c. C3H10: IR absorptions at 3108-2875, 1606, and 1496 cm H NMR spectrum 3 H 5 H 2H 0. 8. 7. 6. 4. 3 ppm GIAY Focus 2. 3. 2. COarrow_forwardGiven here are 1H-NMR and 13C-NMR spectral data for nine compounds. Each compound shows strong absorption between 1720 and 1700 cm-1 and strong, broad absorption over the region 2500–3300 cm-1. Propose a structural formula for each compound.arrow_forwardPropose a structure for each of the following two isomers with formula C6H14 given their 1H-NMR spectra. Isomer A: δ = 0.84 (d, 12 H), 1.39 (septet, 2H) ppm Isomer B: δ = 0.84 (t, 3 H), 0.86 (t, 9H), 1.22 (q, 2H) ppmarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License