
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
9th Edition
ISBN: 9780136781776
Author: Wade
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 16, Problem 16.31SP
In Kekulé’s time cyclohexane was unknown, and there was no proof that benzene must be a six-membered ring. Determination of the structure relied largely on the known numbers of monosubstituted and disubstituted benzenes, together with the knowledge that benzene did not react similarly to a normal
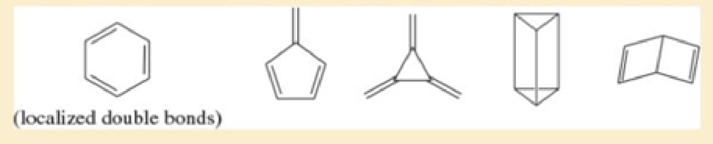
- a. Show where the six hydrogen atoms are in each structure.
- b. For each structure, draw all the possible monobrominated derivatives (C6H5Br) that would result from randomly substituting one hydrogen with a bromine Benzene was known to have only one monobromo derivative.
- c. For each of the structures that had only one monobromo derivative in part (b), draw all the possible dibromo derivatives. Benzene was known to have three dibromo derivatives, but resonance theory was unknown at the time.
- d. Determine which structure was most consistent with what was known about benzene at that time. Benzene gives one monobrominated derivative and three dibrominated derivatives, and it gives negative chemical tests for an alkene.
- e. The structure that was considered the most likely structure for benzene is called Ladenburg benzene, after the chemist who proposed it. What factors would make Ladenburg benzene relatively unstable, in contrast with the stability observed with real benzene?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Below is the structure for beta-carotene, a pigment found in plants and vegetables that give them
their color. Determine how many alkenes are properly trans in the structure.
The chemical structure of propane (C3H3) is provided below. Which of the
following isomers is propane able to form? Briefly justify your answer (2-
3 sentences)
A. Structural isomer
B. Cis-trans isomer
C. Enantiomer
D. All of the above
E. None of the above
н
ннн
H-C-C-C-H
H.
ннн
H.
Draw a resonance structure, complete with all formal charges and lone (unshared) electron pairs, that shows the resonance interaction of the chloro with the para position in chlorobenzene.
0
chlorobenzene
• You do not have to consider stereochemistry.
• Include all valence lone pairs in your answer.
In cases where there is more than one answer, just draw one.
.
99-85
On [F
ChemDoodleⓇ
Chapter 16 Solutions
EP ORGANIC CHEMISTRY -MOD.MASTERING 18W
Ch. 16.2 - Prob. 16.1PCh. 16.2 - Prob. 16.2PCh. 16.2 - a. Draw the resonance forms of benzene,...Ch. 16.2 - Show the product of the Diels-Alder dimerization...Ch. 16.4 - Prob. 16.5PCh. 16.6 - Make a model of cyclooctatetraene in the tub...Ch. 16.6 - Prob. 16.7PCh. 16.6 - Prob. 16.8PCh. 16.7 - Prob. 16.9PCh. 16.8A - a. Draw the molecular orbitals for the...
Ch. 16.8A - Repeat Problem16-10 for the cyclopentadienyl ions....Ch. 16.8C - Explain why each compound or ion should be...Ch. 16.8C - The following hydrocarbon has an unusually large...Ch. 16.8C - Prob. 16.14PCh. 16.8C - Prob. 16.15PCh. 16.9B - Prob. 16.16PCh. 16.9C - Show which of the nitrogen atoms in purine are...Ch. 16.9C - The proton NMR spectrum of 2-pyridone gives the...Ch. 16.9D - Prob. 16.19PCh. 16.9D - Prob. 16.20PCh. 16.10 - Prob. 16.21PCh. 16.12 - Ciprofloxacin is a member of the fluoroquinolone...Ch. 16.13 - Draw and name all the chlorinated benzenes having...Ch. 16.13 - Name the following compounds:Ch. 16.15 - The UV spectrum of 1-phenylprop-2-en-1-ol shows an...Ch. 16 - Prob. 16.26SPCh. 16 - Name the following compounds:Ch. 16 - Draw and name all the methyl, dimethyl, and...Ch. 16 - Four pairs of compounds are shown. In each pair,...Ch. 16 - One of the following hydrocarbons is much more...Ch. 16 - In Kekuls time cyclohexane was unknown, and there...Ch. 16 - Prob. 16.32SPCh. 16 - Azulene is a deep-blue hydrocarbon with resonance...Ch. 16 - Prob. 16.34SPCh. 16 - Prob. 16.35SPCh. 16 - Prob. 16.36SPCh. 16 - Prob. 16.37SPCh. 16 - Prob. 16.38SPCh. 16 - Prob. 16.39SPCh. 16 - Biphenyl has the following structure. a. Is...Ch. 16 - Anions of hydrocarbons are rare, and dianions of...Ch. 16 - How would you convert the following compounds to...Ch. 16 - Prob. 16.43SPCh. 16 - Prob. 16.44SPCh. 16 - A student found an old bottle labeled thymol on...Ch. 16 - Prob. 16.46SPCh. 16 - Prob. 16.47SPCh. 16 - Prob. 16.48SPCh. 16 - The proton NMR chemical shifts of the hydrogens in...Ch. 16 - Prob. 16.50SPCh. 16 - NMR has been used to probe many molecular...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Compounds X and Y both have the formula C7H₁4. Both X and Y react with one molar equivalent of hydrogen in the presence of a palladium catalyst to form 2-methylhexane. The heat of hydrogenation of X is greater than that of Y. Both X and Y react with HCI to give the same single C7H15Cl compound as the major product. What is the structure of X? • In cases where there is more than one answer, just draw one. 23 ▾ Sn [F ChemDoodleⓇ 146arrow_forwardDraw a full Lewis structure for each following compound. Include lone pairs andnon-zero formal charges as necessary.a. HCN, hydrogen cyanideb. Si(OH)4, orthosilicic acid c. and d. are in photo attached. Look at cyclobutan-2,2-diol from d. Write the hybridizaOon of eitheroxygen atom (sp, sp2, or sp3). In Si(OH)4 (from question b.), write the hybridization of the silicon atom. Write compound in in this previous question that should exhibit a strong, sharp peak at 1712cm–1 in its IR spectrum.arrow_forwardNew hydrocarbon compounds K, L, M and N have been formed from an organic reactions in Dr. Meredith laboratory. She found that compound K and L with molecular formula (C;H14) are unsaturated hydrocarbons with positional isomerism. Meanwhile, the reaction of hydrocarbon from compound M produces carbon dioxide and water when burns in plentiful oxygen. Another observation showed compound N produces three products X, Y and Z when react with chlorine gas in the presence of UV light. Identify the name of unsaturated hydrocarbon compound K and L and draw their possible condensed and skeletal structures. Suggest the IUPAC nomenclature name for compound M and write the complete chemical reaction of compound M when burns in plentiful oxygen. Show the complete mechanism reaction that occur for the formation of products X, Y and Z from compound N.arrow_forward
- a. b. C. a. b. XV. Refer to structure below to answer the following questions: XVI. Which of the label bonds is trans to bond b? с Which of the label bonds in the following structure are equatorial bonds? a (CH3)3C b Which bonds have a 1,3 - diaxial interaction with each other? CH3 For each substituted cyclohexane below, draw its ring-flip isomer. Show all the 1,3-diaxial interactions and the gauche interactions if they occur and predict (without any calculations) the most stable conformer. CH3 d CH3arrow_forward8. Beta-carotene, CaoHse, is found in carrots and many other veggies. Beta-carotene reacts with excess hydrogen over a Pd/C catalyst to give CaoHra. How many rings are in beta-carotene? How many double bonds are in beta-carotene? Show how you got your answers.arrow_forwardQ5-A- Define Covalent bond with example? B- Choose the correct answer (4 only): 1-The molecular formula of ethane is: A.CH4 В. С2Н6 С. СзНя D.C4H10 2- Which of the following is not an alkene? A. Propane B. Butene С. C:H12 D. C4H10 3-As compared to its parent alkane, an alkyl radical contains one A. less carbon B. less hydrogen C. more carbon D. more hydrogen 4- Almost 95% of compounds are of carbon because they can form A.single bonds B. double bonds C.Triple bonds D.Multiple bond 5- The general formula for alkynes is : A. CnH2n-2 B. CnH2n C. CnH2n-2 D. nH2n-1arrow_forward
- 9. There are several possible forms of a trisubstituted cyclohexane with the formula C10H200. I have drawn four of them. From these, which one do you think is most commonly naturally occurring, and why? Which is least commonly occurring and why? HO HO" НО HOarrow_forward3. Please draw the most important (stable) resonance form of the carbonation that mediates the following reaction: OH H3O+ O 4. Draw the structure of the alkene isomer (with the molecular formula of C5H10) that is most reactive one in the addition reaction with Br₂.arrow_forwardThe enthalpy for hydrogenation of different isomers of butene is shown in the following figure. Which statement below best interprets this figure? Hy I3D H2 7k mot 5kJ mot + H2 AH-120 kJ mot AH"-127 kJ mot AH-115 kJ mot © GMU 2020 Stability and hydrogenation enthalpies are not correlated. Butene II is less stable than butene I by 7 kJ/mol. O Butene Il is 7 kJ/mol more stable than I and 5 kJ/mol more stable than III. Butene II is 5 kJ/mol less atable than butene IlI. Enthalpyarrow_forward
- Draw a structure for 1,1-dichloro-3-propoxycyclopentane. • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • In cases where there is more than one answer, just draw one. opy astearrow_forward4. Write the condensed formula of the following structures. H H e. 3. N-O hydroxylamine Н H-C Н с ΗΤῊ Н 2-methylpropan-2-ol ΗΙ I 1 Н Н -С-СЕС-С 1 1 Н HIH C I нн С- НА 'H H 6-bromo-4-methylhex-2-yne Br Г C11 b. d. Н HIH с н-с-с-с-Н C ΗΤῊ H neopentane нн H Н H H нн ethane-1,2-diolarrow_forwardWhen exactly 1 mole of methane is mixed with exactly 1 mole of chlorine and light is shone on the mixture, a chlorination reaction occurs. The products are found to contain substantial amounts of di-, tri-, and tetrachloromethane, as well as unreacted methane. How would you run this reaction to get a good conversion of methane to CH3Cl? Of methane to CCl4?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY